Is it the right time to address the issue of over-the-counter (OTC) and self-medication in Ayurveda?
Badal Robin*. PhD Scholar, Second Year- Department of Rasashastra & Bhaishajya Kalpana, All India Institute of Ayurveda, Sarita Vihar, New Delhi -110076, India. Email: robin.baadal2608@gmail.com
Patil Giramalla. Assistant Professor- Department of Rasashastra & Bhaishajya Kalpana, All India Institute of Ayurveda, Sarita Vihar, New Delhi -110076, India. Email: girishpatil. patil2@gmail.com
Yadav Pramod. Associate Professor- Department of Rasashastra & Bhaishajya Kalpana, All India Institute
of Ayurveda, Sarita Vihar, New Delhi -110076, India. Email: drpramod88@gmail.com
Prajapati PK. Vice Chancellor-Dr. Sarvepalli Radhakrishnan Rajasthan Ayurved University (DSSRAU), Jodhpur-342037, India. Email: prajapati.pradeep1@gmail.com
DOI: 10.18549/PharmPract.2025.3.3139
Abstract
Background: Ayurveda, a trusted holistic healthcare tradition, is widely embraced for its therapeutic benefits. In India, the practice of self-medication with over-the-counter (OTC) Ayurveda medicines is common, lacking specific regulations akin to the historical Vaidya Parampara era. This study aimed to explore the necessity of regulations for OTC Ayurveda medicines, considering safety and quality concerns. Methods: An extensive literature review was conducted across online databases, including Google Scholar, PubMed, and others, focusing on OTC Ayurveda medicines. Additionally, a pilot survey involving 106 patients and 34 medical stores was conducted to assess attitudes toward OTC Ayurveda medicines utilizing a structured questionnaire. Results: This study revealed a dearth of specific regulations for OTC Ayurveda medicines, posing challenges in distinguishing between prescription and nonprescription drugs. Both pharmacists and patients exhibited varied attitudes toward OTC Ayurveda medicines, with concerns spanning safety, quality, accessibility, and awareness. Discussion: This article examines the necessity of regulations for OTC Ayurveda medicines, considering their advantages and disadvantages across different stakeholders. The discussion emphasized the critical role of regulations in ensuring patient safety and quality assurance and maintaining the integrity of Ayurveda’s holistic principles. Conclusion: In conclusion, this study underscores the urgent need for regulatory measures governing OTC Ayurveda medicines to ensure patient safety and quality control. By addressing the complexities and challenges associated with the current regulatory landscape, appropriate regulations can safeguard public health while preserving the essence of Ayurveda.
Keywords: Over the counter, Self-medication, Ethics code, Ayurvedic medicine.
INTRODUCTION
The drug applied wrongly acts as a poison, while it is rightly administered as nectar. The right drug should also be used cautiously at prescribed times.1 Ayurveda, with its holistic approach to health and well-being, extends its influence from household practices (Gharelu Upchar) to healthcare facilities such as hospital inpatient departments (IPDs) and wellness centers. Self-medication, defined by the World Health Organization (WHO) as the use of drugs to treat self-diagnosed disorders or symptoms, is a widespread practice in India.2 The prevalence of self-medication in the Indian population varies significantly, with rates ranging from 8.3% to 93%.3 Low-income families who cannot afford health services exhibit a greater tendency toward self-medication.4 Over-the-counter (OTC) or nonprescription medicines, authorized for sale by pharmacists without the need for a prescription from a registered medical practitioner, are readily available in drugstores, convenience stores, and supermarkets.5 However, certain medications listed under different schedules in the Drugs and Cosmetics Act and the Drugs and Cosmetics Rule have restrictions on their sale.6 7 Medications listed in Schedules H, H1, and X should carry a label stating that these drugs are to be sold by retail only on the prescription of a registered medical practitioner other than these all are considered nonprescription medicines.
During the zenith of the Vaidya Parampara (legacy) in Ayurveda, ancient scholars personally formulated and dispensed medicines to patients based on individual health conditions. This approach meant that there were no regulations governing over-the-counter (OTC) medications during that time. However, practitioners understood that an unfamiliar drug could be as perilous as poison, a weapon, fire, or a thunderbolt. Conversely, a known substance was regarded as nectar.8 They also knew that drug reactions could vary widely post-administration, ranging from excessive reactions to mild reactions or, in some cases, no observable effects. Ensuring the administration of the correct drug at the right dosage and at the right time minimizes the likelihood of complications.9
Currently, the Drugs and Cosmetics Act of 1940 and Rules of 1945 do not encompass specific regulations about the sale of over-the-counter (OTC) Ayurveda medicines, and there is no demarcation between which one is sold under prescription and which one is not. Ayurvedic medicines not only are available in pharmacies but also have a prominent presence, with almost 100% availability in grocery stores, malls, and supermarkets. Over-the-counter (OTC) medicines have both positive and accessible and affordable healthcare options contribute to the overall improvement of public health. The integration of the third Sustainable Development Goal (SDG), “Good Health and Well-being”, into the OTC drug sector involves ensuring that accessible and affordable healthcare options contribute to the overall improvement of public health. To assess the attitudes of commercial pharmacists and patients, a comprehensive survey encompassing both patients’ experiences and insights from nearby medical stores was administered to 106 patients and 34 medical stores. Patient experiences and perspectives from the pharmacists were recorded and analyzed. In this article, we will discuss the need for rules and regulations for OTC medicines, their advantages and disadvantages, and their impact on patients, consumers, and the industry and outcome of survey studies.
METHODS
Why common people accept without prescription
Myth
There is a common misconception that Ayurveda is entirely devoid of side effects/adverse effects. However, it is important to acknowledge that improper use or lack of professional guidance in Ayurvedic treatments may result in adverse effects (Figure 1).
Social Media
In the current scenario, social media and health/wellness influencers play a pivotal role as primary sources of information, whether accurate or not. Many individuals tend to unquestioningly follow these influencers, and influencers do not even have degree holders or any professional experience. By utilizing social media platforms, these influencers endorse and market Ayurvedic products without holding relevant degrees or clinical expertise. This raises dual concerns: the influencers’ absence of clinical experience and the second is that they can sell substandard or counterfeit Ayurvedic medicines. 10 11
E-Market
All medicines are easily accessible without a prescription and without any restriction on quantity. Rasa aushadhies (metal and mineral preparation) and Schedule E-1 (poisonous substances) D&C Act drugs and formulations containing them are also easily available on the e-market.
Family/relative self-medication experience/recommendation/ WOM
When someone in the family or among friend’s experiences being cured with a specific medication, they often share their feedback or word of mouth (WOM) and recommend taking the same medicine without undergoing a proper diagnosis or consulting a doctor.
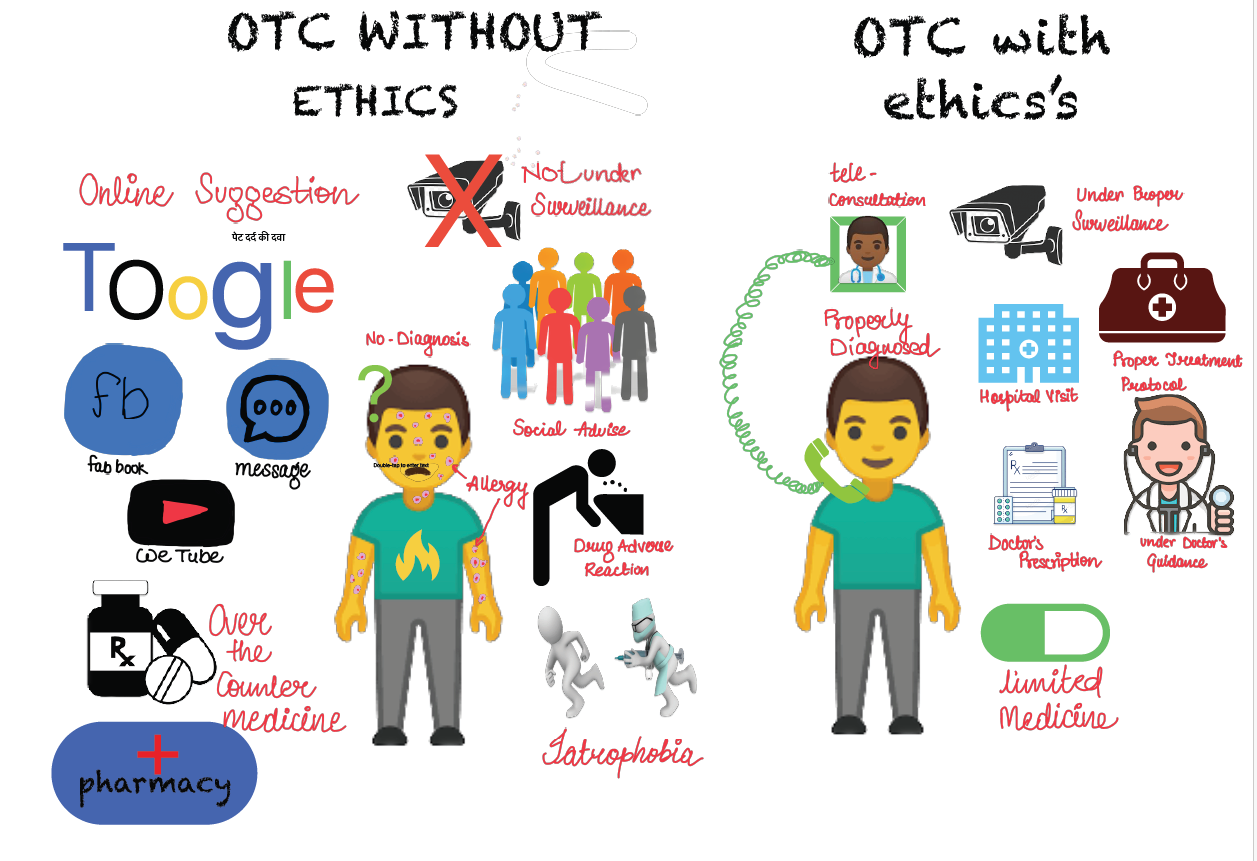
Intentional Misuse/Abuse
In the context of the social stigma associated with individuals facing diseases of Vigor and vitality, many patients often opt for self-medication due to Iatrophobia. Individuals may misuse or abuse medications, primarily for personal reasons such as addiction. In the context of self-medication, patients commonly resort to the use of substances aimed at boosting immunity, promoting rejuvenation, increasing muscle gain, increasing hair growth, increasing height, or serving prophylactically.
The names of ayurveda/herbal medicine/natural/ nutraceutical
Ayurvedic medicines are sold abundantly, akin to a popular commodity in high demand.
ADVANTAGES AND DISADVANTAGES AT FOUR-LEVEL/ STAKEHOLDERS
Patient/consumer perspective
In terms of advantages, it is cost-effective, readily available, and does not require consultations. However, on the downside, patients often lack knowledge about crucial aspects such as dosage, duration, frequency, Anupana (vehicle), and disease conditions (stages); the potential hazards include the development of drug resistance, adverse drug reactions, the possibility of interactions with other drugs or food and concomitant drug administration. A published study involving 58 patients revealed a 74% incidence of high-risk drug‒ drug interactions (DDIs).12 Notably, 52% of the total DDIs identified were attributed to interactions between prescribed medications and over-the-counter (OTC) or herbal products.
Doctors/physicians’ perspectives
At the level of healthcare professionals, there are minimal benefits such as less crowding at the healthcare facility, but the potential for harm is significant. Professional losses may include a doctor’s comprehensive understanding of diagnosis, prognosis, the stage of the disease, and treatment protocols, including Aushadh Pana Niyam (medication administration guidelines).13 Particularly in Ayurveda, considering the individual Prakriti (body constitution) is crucial, as certain medications are explicitly contraindicated for specific Prakriti. When a patient engages in prolonged self-intervention, complications may arise, leading to the presentation of a complicated case when seeking professional assistance.
Pharma industry/manufacturer perspective
In pharma, the benefits are greater, revenue generation is greater, no medical representative is needed, and a direct approach to consumers and online pharmacies is thriving in this environment. The primary concern lies in assigning responsibility when a patient makes an incorrect decision regarding their chosen drug or if there is an error on the part of the pharmacist. In such instances, the blame is often directed solely at the pharmaceutical company.
Rule regulation authority
The sole advantage is a reduction in the healthcare burden. However, there are numerous drawbacks, including the defamation of traditional treatment practices (pathy) and the challenge for authorities to discern the extent of the issues at hand. In a reported study that analyze randomly collected common over-the-counter Ayurvedic preparations from licensed Ayurvedic shops, heavy metals were detected at concentrations greater than the permissible limits for all formulations, according to the permissible limits set by the Food and Agriculture Organization/World Health Organization (FAO/WHO) for herbal medicines.14 This raises concerns about the acceptability of OTC products and herbal medicines in general.
WHO GUIDELINES MENTIONED THE RISK FACTORS OF OVERTHE- COUNTER MEDICATION15
Incorrect Self-Diagnosis: The process of self-medication commonly begins with self-diagnosis, which carries a high risk of inaccuracy.
Misguided Therapy Choices: Even with a correct diagnosis, there is a likelihood of making incorrect therapeutic choices.
Unawareness of Special Groups: Consumers may not be aware of their inclusion in special groups with potential pharmacological side effects, such as pregnancy, immunocompromised conditions, or advanced age.
Ignorance of Contraindications: Individuals without medical expertise may be unaware of drug contraindications, warnings, and precautions, potentially resulting in severe adverse drug reactions.
Risk of Double Medication: The risk of unintentional double medication exists, especially if a patient is already taking the same active substance under a different name.
Harmful Drug Interactions: The concomitant use of self-medicated drugs with other medications may lead to harmful drug interactions.
Incorrect Route of Administration: There is a potential for using medications via the wrong route.
Self-Dosing Risks: Self-dosing, especially over prolonged periods, increases the risk of habitual body activity, dependency, and future drug abuse. For example, senna (Cassia angustifolia) is a common ingredient in many over-the counter laxatives, but it is advised for short-term usage (less than one week).16 Prolonged use or misuse may result in a “cathartic” colon, manifesting as diarrhea, abdominal cramps, electrolyte imbalance, weight loss, and darkened pigmentation of the colonic mucosa.
Shortage for Those in Need: Self-medication contributes to the scarcity of drugs for individuals with genuine medical needs for other diseases.
ADDRESSING THE CHALLENGES ASSOCIATED WITH OTC/SELFMEDICATION REQUIRES A MULTIFACETED APPROACH
Public Education: Implement comprehensive public awareness campaigns to educate individuals about the risks and consequences of self-medication The importance of consulting healthcare professionals for accurate diagnosis and appropriate treatment should be emphasized. For example, for public awareness, the Ministry of Ayush issued a Technical Dossier on Guduchi (Tinospora cordifolia). 17
Enhanced Regulatory Measures: Strengthen regulatory frameworks to monitor and control the sale of medications, particularly those prone to misuse. Stringent guidelines for over-the-counter drug dispensing should be enforced. For example, in a published study, Ayurveda medicine samples were collected from markets, supermarkets, and convenience stores, revealing that 14 out of 70 Ayurvedic formulations contained heavy metals.18 In another study, samples acquired through online purchases indicated that one-fifth of both USmanufactured and Indian-manufactured products contained detectable levels of lead, mercury, or arsenic. If rules and regulations had been strengthened, then defamation of the pathy would not have happened.19
Behind-the-counter medicine: Pharmacists can play a pivotal role in providing information on proper medication usage, potential side effects, and interactions. Educate the pharmacist and should possess four essential qualities in terms of OTC:20 Buddhiman (persuade the patients to produce prescriptions), Daksha (He should be very much attentive to the latest developments and regulations), Anurakta (affection and attachment toward the patient), and Shuchi (professional purity and ethical practice). and encourage the active involvement of pharmacists in guiding consumers. In an Indian study, over 90% of interviewed qualified pharmacists were familiar with OTC drugs.21 When approached for OTC drug purchases, 96.5% of respondents inquired about patients’ complaints, but only 51% provided counseling on usage instructions.
Telemedicine Services: Promotion of telemedicine services to facilitate remote consultations with healthcare professionals This can offer accessible and timely advice on health concerns, reducing the reliance on self-diagnosis.
Community Health Initiatives: Implement community health initiatives aimed at improving health literacy Community leaders, schools, and local healthcare providers should collaborate to share precise information about health practices and the repercussions of self-medication.
Technology Integration: Leverage technology to create userfriendly platforms that provide reliable health information. Encourage the use of reputable health apps and websites for self-assessment, with clear disclaimers promoting professional consultation for accurate diagnosis. For example, the “Ayush Sanjivani” mobile application, introduced by the Ministry of AYUSH, offers insights into the strategies adopted by the public to boost immunity and maintain well-being during the challenging COVID-19 situation.
Collaboration with Stakeholders: Collaboration among pharmaceutical companies, healthcare professionals, regulatory bodies, and the public to create a unified effort to promote responsible medication use
Pharmacovigilance impact: By cultivating a reporting culture, conducting robust risk assessments, and advocating for transparency, pharmacovigilance can play a pivotal role in promoting safer self-medication choices and enhancing overall public health protection.
RESULTS
Observations of the survey study
In the survey study, a questionnaire of 9 assorted questions was administered to 106 patients, whereas a questionnaire of 10 questions was administered to 34 pharmacists. The findings of the survey are shown in Table 1 and Table 2. The enrolled patients were recruited from the outpatient department (OPD) of Ayurveda Hospital, spanning an age range of 18 to 83 years. Among them, there were 62 males and 44 females. Out of the 106 patients, 26 responded negatively to the question “Haveyou ever used Ayurvedic medicine before?” The remaining 80 patients participated in the full questionnaire. On the basis of the responses, most of the patients sought OTC drugs pertaining to the GI tract and musculoskeletal ailments. Generally, these ailments have become increasingly prevalent due to the adoption of Western lifestyles, leading to the development of gastrointestinal and musculoskeletal disorders, although most of the patients are unaware of the concept of OTC from the Ayurveda perspective. Encouragingly, most of the patients usually approached a healthcare provider to gather information about Ayurveda drugs (40%). Furthermore, nearly 20% of the participants relied on OTC Ayurveda drugs to meet their day-to-day health care needs. Moreover, Ayurveda drugs are easy to obtain from OTC for most patients. Thus, Ayurvedic medicines are easily available on an OTC basis, and patients are least aware of any regulatory framework guiding their sale.
The observations from a chemist’s perspective are worthy of note. The surveyed chemists ran allopathic pharmacies with small sections dedicated to Ayurveda drugs. From the survey, it is evident that chemists sell Ayurveda drugs without any prescription. Most of the customers were approached for the purpose of Vajikarana (Aphrodisiac), followed by GI problems. This shows the attitude and understanding of the general public toward Ayurveda drugs. It is still seen as the most accepted medium for elevating vigor and vitality rather than a therapypoint of view was different due to social constraints, dogma, and taboos related to discussions pertaining to sexual health and well-being. The majority of chemists were not aware of potential interactions and ADRs related to Ayurveda drugs, and neither were they bothered to explain the posology and proper drug administration of Ayurveda drugs to their customers. They also despised regarding the formulation of any regulatory framework to countercheck OTC Ayurvedic medicines due to possible monetary aspects related to profit and sales.
DISCUSSION
Ways forward/remarks
A Vaidyah (Physician) contributes significantly to the holistic success of the pādacatuṣṭayam (four components of medical practice) because while fixing a dose, a knowledgeable physician should carefully consider multiple factors, such as the stage of the disease, the patient’s age, the overall condition of the disease, and other relevant health parameters. The effectiveness of a treatment is contingent upon the accurate assessment of various factors, including the appropriate timing and dosage.22 In Ayurveda, it is imperative to establish clear rules and regulations governing the classification of Ayurvedic products, distinguishing between those products that can be sold with or without a prescription or can be classified (a) Prescription drugs, (b) pharmacist-only drugs, and (c) over-the-counter medicines. Similar to the modern medicine paradigm, Ayurveda should adopt a comprehensive “Rule Book” outlining the monographs of over-the-counter (OTC) drugs.23 To streamline this process, a curated list, including Ashwagandha (Withenia somnifera), Amla (Emblica officinalis Gaertn. ), Haridra (Curcuma longa Linn.), Vasa (Adhatoda vasica Nees. ), and Tulsi (Ocimum tenuiflorum L.), can be developed to address common ailments. Furthermore, it is crucial to exercise caution related to Rasa aushadhies and exclude Schedule E1 drugs from the OTC list. Additionally, there should be limitations on alcohol-based Ayurveda drugs, particularly those falling under the category of Asava-Aristha (Alcoholic fermented preparation).
In labeling OTC Ayurvedic drugs, it is essential to include a “Drug Fact” section providing information on active ingredients, dosage, duration, frequency, and Anupana (vehicle). This section should also highlight any interactions, specifying which drugs or foods should be avoided when consuming the particular formulation, incorporating visual representations that can be read easily and understood by a layperson. This approach ensures that consumers have clear and comprehensive information to make informed decisions about their health and well-being.
CONCLUSION
As a result, there is an urgent need to monitor and manage acceptable self-medication and OTC drug practices through the use of strong laws and the involvement of healthcare experts and policymakers. The popularity of Ayurveda has steadily grown since COVID-19, and with its increasing global presence, there has been heightened scrutiny of its product effects and side effects. This underscores the significance of addressing the issues associated with over-the-counter (OTC) products at this juncture.
CONFLICT OF INTEREST
None.
ACKNOWLEDGMENTS
We sincerely thank the All India Institute of Ayurveda, New Delhi, for providing financial support for this work. We also express our heartfelt gratitude to Dr. Niladri Bhattacharjya and Dr. Manish Kumar Saini for their invaluable contributions to the conceptualization and drafting of this paper. Additionally, we are deeply grateful to Dr. Payal Maheshwari for her efforts in creating the pictorial representation featured in this article.
References
1. Acharya YT editor. Charaka Samhita of Agnivesha. 2nd edition. Acharya YT, editor. Vol. chapter 12. Varanasi: Choukhamba orientalia; 2017. 675–675 p.
2. Guidelines for the regulatory assessment of medicinal products for use in self-medication [Internet]. [cited 2025 Feb 4]. Available from: https://iris.who.int/handle/10665/66154
3. Rashid M, Chhabra M, Kashyap A, Undela K, Gudi SK. Prevalence and Predictors of Self-Medication Practices in India: A Systematic Literature Review and Meta-Analysis. Curr Clin Pharmacol [Internet]. 2020 Nov 25 [cited 2025 Feb 4];15(2):90–101. Available from: https://pubmed.ncbi.nlm.nih.gov/31763976/
4. Sisay M, Mengistu G, Edessa D. Epidemiology of self-medication in Ethiopia: A systematic review and meta-analysis of observational studies. BMC Pharmacol Toxicol [Internet]. 2018 Sep 10 [cited 2025 Feb 4];19(1):1–12. Available from: https://bmcpharmacoltoxicol.biomedcentral.com/articles/10.1186/s40360-018-0248-8
5. Verma R, Mohan L, Pandey M, Banarasi B. Evaluation of self medication among professional students in North India: proper statutory drug control must be implemented. 2010;
6. Acts & Rules [Internet]. [cited 2025 Feb 4]. Available from: https://cdsco.gov.in/opencms/opencms/en/Acts-Rules/
7. Marathe PA, Kamat SK, Tripathi RK, Raut SB, Khatri NP. Over-the-counter medicines: Global perspective and Indian scenario. J Postgrad Med [Internet]. 2020 Jan 1 [cited 2025 Feb 4];66(1):28–34. Available from: https://pubmed.ncbi.nlm.nih.gov/31898596/
8. Charak samhita. Charak samhita of Agnivesha. 2nd edition. Acharya YT, editor. Vol. chapter 1 verse 124. Varanasi: Choukhamba orientalia; 2017. 23–23 p.
9. Charak Samhta. Charak samhita of Agnivesha. 2nd ed. Acharya YT, editor. Vol. chapter 6, ver 31. Varanasi: Choukhamba orientalia; 2017. 706–706 p.
10. Regulation and Prequalification [Internet]. [cited 2025 Feb 4]. Available from: https://www.who.int/teams/regulationprequalification/incidents-and-SF/background/definitions
11. World Health Organization steps up action against substandard and counterfeit medicines [Internet]. [cited 2025 Feb 4]. Available from: https://www.who.int/news/item/11-11-2003-world-health-organization-steps-up-action-againstsubstandard-and-counterfeit-medicines
12. Yoon SL, Schaffer SD. Herbal, prescribed, and over-the-counter drug use in older women: prevalence of drug interactions. Geriatr Nurs [Internet]. 2006 Mar [cited 2025 Feb 4];27(2):118–29. Available from: https://pubmed.ncbi.nlm.nih.gov/16638483/
13. Vangasena. Vangsena Samhita. 1st ed. Pandit HPT, editor. Vol. vers 85-86. Varanasi: Choukhamba orientalia; 2009. 21–21 p.
14. Bhalla A, Pannu AK. Are Ayurvedic medications store house of heavy metals? Toxicol Res (Camb) [Internet]. 2022 Feb 1 [cited 2025 Feb 4];11(1):179–83. Available from: https://pubmed.ncbi.nlm.nih.gov/35237422/
15. WHO guidelines for the regulatory assessment of medicinal products for use in self-medication : general information. WHO drug information 2000 ; 14(1) : 18-26 [Internet]. 2000 [cited 2025 Feb 4]; Available from: https://iris.who.int/handle/10665/57624
16. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet] - PubMed [Internet]. [cited 2025 Feb 4]. Available from: https://pubmed.ncbi.nlm.nih.gov/31643176/
17. Guduchi (Tinospora cordifolia).
18. Saper RB, Kales SN, Paquin J, Burns MJ, Eisenberg DM, Davis RB, et al. Heavy metal content of ayurvedic herbal medicine products. JAMA [Internet]. 2004 Dec 15 [cited 2025 Feb 4];292(23):2868–73. Available from: https://pubmed.ncbi.nlm.nih.gov/15598918/
19. Saper RB, Phillips RS, Sehgal A, Khouri N, Davis RB, Paquin J, et al. Lead, mercury, and arsenic in US- and Indian-manufactured Ayurvedic medicines sold via the Internet. JAMA [Internet]. 2008 Aug 27 [cited 2025 Feb 4];300(8):915–23. Available from: https://pubmed.ncbi.nlm.nih.gov/18728265/
20. Charak. Charak samhita . Dr. Brahmanand Tripathi and Dr. Ganga Sahay Pandey, editor. Vol. chapter 9, verse 8. Chaukhamba Surbharti Prakashan; 2007. 209–209 p.
21. Ravichandran A, Basavareddy A. Perception of pharmacists regarding over-the-counter medication: A survey. Indian J Pharmacol [Internet]. 2016 Nov 1 [cited 2025 Feb 4];48(6):729. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC5155479/
22. Agnivesha. Charak Samhita of Agnivesha. 2nd ed. Vol. chapter 2, verse 6. Varanasi: Choukhamba orientalia; 2017. 25–25 p.
23. Babar ZUD. Ten recommendations to improve pharmacy practice in low and middle-income countries (LMICs). J Pharm Policy Pract [Internet]. 2021 Dec 1 [cited 2025 Feb 4];14(1):1–5. Available from: https://joppp.biomedcentral.com/articles/10.1186/s40545-020-00288-2
