The burden cost of adverse drug reaction related to admission at Chaophrayayommarat hospital
Monton Tanabodee*. MD, Division of dermatology, Department of Internal Medicine, Chaophrayayommarat hospital. montontana@hotmail.com
Nirun Jangkong. Pharm, Department of Pharmacy, Chaophrayayommarat hospital. pomoq73@gmail.com
DOI: 10.18549/PharmPract.2026.3.3634
Abstract
Background: Adverse Drug Reaction-related Hospital Admissions (ADRA) represent a significant impact on clinical problem and healthcare expenditure. This study aimed to determine the clinical manifestations, prevalence and cost burden from ADRA in the context of Chaophrayayommarat hospital, regional hospital level, Thailand. Methods: An observational study of adult patients who unplanned hospital admissions to Chaophrayayommarat hospital caused by adverse drug reactions (ADRs) during May 2024 – March 2025. The prevalence of ADRs, their economic burden from the hospital perspective, severity, and preventability were assessed using standard criteria. Results: Out of 23,247 hospital admissions, 217 ADRA have been identified. The incidence of ADRA was 0.93% (95% CI 0.93±0.001). Antimicrobial agents related ADRs, Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Anti-diabetic were the commonest ADRs seen accounting for 21.18, 14.28, and 13.36 events respectively out of the 217 events. Out of 217 events, 144 (66.35%) were identified as the potential preventability of ADRA. The total cost for 217 patients with ADRA admitted to Chaophrayayommarat hospitals was estimated at 1,555,206 Bath. The mean total cost per patient with ADRA was estimated at 7215 ± 4193 Bath. Conclusion: In addition to the increase in the number of expensive drugs, the ageing population and polypharmacy, the economic impact of ADRA weighs on healthcare spending. It should be emphasized that in most ADRA, medications were contributory reason of hospital admissions and that benefits and risks have to be carefully balanced.
Keywords: adverse drug reactions, health expenditure
INTRODUCTION
The World Health Organization (WHO) defines Rational Drug Use (RDU) as patients receiving medication appropriate to their needs, in correct doses, for the right duration, and at the lowest cost. For RDU in Thailand, The Ministry of Public Health (MOPH) plays a crucial role in ensuring RDU through regulatory policies and guidelines with a focus on safety, especially adverse drug reaction (ADRs) is a key area of public health concern1. ADRs is a health problem occurring from medication intervention. It includes adverse drug reaction, overdose and any noxious and unintended response to a drug and occurs at doses used for prophylaxis, diagnosis, or therapy in humans, excluding failure to accomplish the intended objective2,3. ADRs is one of the major causes of patient related morbidity and mortality worldwide and place a significant economic burden4,5.
An epidemiological study of adverse drug reaction-related hospital admissions (ADRA) showed 2.5-10.6% of hospital admissions in Europe, 5.7-18.8% in Australia, and 4.2-30% in the USA and Canada5. In USA, the estimated expenditure for ADRs management was 30.1 – 130 billion US dollars per year5,6 The estimated direct hospital costs of ADRs in Australia was 4.83 – 9.00 billion Australian dollars annually and half of them may be preventable7. As a part of pharmacovigilance, ADRs monitoring system can detect the signal for adverse events before clinically manifestation but, ADRs reporting is voluntary and was estimated that only 6-10% of all ADRs are reported8-10. In the context of the increasing complexity of therapy and an ageing population exposed to the risk of multiple chronic diseases and polypharmacy, ADRA should be remained as a major public health issue with a significant impact on healthcare expenditure.
In Thailand, since 1984, ADRs reporting is under pharmacovigilance center, ‘Health Product Vigilance Center (HPVC)’, an organization that has responsibility to collect and evaluate the reported ADRs from healthcare professionals, approximately 89% of ADRs were reported from hospitals10. As compared to other countries, incidence of ADRs among hospitalized patients in Thailand was also high ranging between 1.7% to 22.6 % depending on methods, study settings and also study population11. It should be noted that all previous studies conducted in Thailand to examine prevalence of ADRs among hospitalized patients were conducted using spontaneous reporting system12-16. Few studies evaluate ADRA, the aims of our study are to examine incidence and characteristics of ADRA through the emergency department at Chaophrayayommarat hospital.
METHODS
Study Design and Setting
This observational study examined hospital admissions to Chaophrayayommarat hospital via emergency department in order to identify those which are suffered from ADRs. The investigator visited study wards daily and reviewed patients’ drug charts, medical and nursing notes for evidence of an ADRs. A suspected marker was used to screen the medical records for
ADRs17-18. Markers of ADRs (e.g., laboratory results, sign and symptoms) were identified through patient notes and discussion with the treating physician and affected patients. Efforts were made to ensure complete work-up during admission in all cases to make proper assessment of ADRs. Suspected ADRs were classified in terms of causality by using Naranjo’s algorithm19. ADRs were also classified as either type A or type B according to the system introduced by Rawlins and Thompson in 197720. For preventable adverse drug reactions (pADRs) refers to classifying as avoidable due to errors like incorrect dosage, ignoring warnings (allergies, drug interactions), inappropriate prescribing (wrong drug, dose, route), patient noncompliance to or without professional guidance, and common side effects itself. As preventability assessment is no specified tool, in essence, the root cause of ADRs helps distinguish between errors that should have been detected (preventable) and those that were truly unavoidable, guiding targeted safety initiatives. Pharmacists work to eliminate the errors and inappropriate regimen that led to preventable ADRs.
Severity of ADRs was assessed using Hartwig severity scale21. Data on the reported ADRs were evaluated to understand the pattern of the ADRs with respect to patient demographics, nature of the reactions, characteristics of the drugs involved, and outcome of the reactions. Causality, severity, preventability, and the presence of predisposing factors for the reactions were assessed. Drugs involved in the ADRs were classified according to the Anatomical Therapeutic Chemical (ATC) classification based on the WHO ATC index 200822. Reactions were further classified depending on the organ system affected using WHO Adverse Reaction Terminology (ART), 200323. Analysis of whether the ADRs directly caused of admission was performed after clinical assessment of the condition in consultation with treating medical team. The costs involved in the management of ADRs were collected, including in the cost of ADRs management, hospital stay, medications, clinical investigation charges, and additional procedures. This analysis used the perspective of the payers in making these estimates, and only direct cost was calculated.
Inclusion and Exclusion Criteria
The study included all patients 20 years and older who were admitted via the department of emergency medicine to any hospital ward of Chaophrayayommarat hospital caused by ADRs, Using the Naranjo’s algorithm, only causality was definite or probable were include. Hospital admissions that took place between May 2024 – March 2025 were included. Visits to the department of emergency medicine without inpatient hospitalization were not included. Hospitalizations for diagnostic or elective surgical procedures for pre-existing conditions, hospitalizations with missing medical records, and hospitalizations taking less than 24 hours were excluded. We also excluded patients who were unable to make an effective interview: unconscious or in any level of coma, acute psychiatric condition, and hearing or speech impairment.
Data Collection
Data were obtained from electronic medical records and entered into a Microsoft Excel database. The collected data included demographic characteristics, medication history, medical history, presenting complaint, admission diagnosis, laboratory values and results of clinical investigations, documented ADRs and information on medication adherence. Medications stated in medication history were counted as active substances.
Ethics Committee Approval
The study was approved by Ethics Committee of Chaophrayayommarat Hospital. Patient informed consent was not required due to the observational design of the study. No personal data that could identify the patients were collected.
Sample Size Calculation
Sample Size We used finite population mean formula from n4Studies application24 at a confidence level of 95% and power of 80%, prevalence from the previous study in a similar setting,25 the minimum sample size required was 196 patients. We added 10% for possible incomplete patient files, and too early discharge of the patients, resulting in a target sample size of 216 Thus, achieving a 2-sided margin of error of less than 5%.
Data Analysis
Descriptive statistics were employed to analyze the frequency and percentages of causality, types, mechanism, severity, and preventability of ADRs as well as the medications implicated in the suspected ADRs. Statistical Analysis Frequencies with percentage were used to summarize gender, diagnosis, number of drugs taken, length of stay, drugs, organ system involved, and severity of ADRs. Mean with 95% confidence interval (CI) was used to summarize age and length of stay. Currency exchange rate, US dollar (USD) to Thai Baht (THB), 1 dollar equal 32.40 baht. Chi-square test was used to find an association between gender and age groups with ADRs. A significance level of p < .05 was considered as statistically significant. All of the statistical analyses were performed using SPSS 25. Categorical variables were expressed as absolute values and percentages. Continuous variables were expressed as medians with interquartile ranges.
RESULT
During study period, 23,247 patients presented to the emergency department (ED). There were 217 with ADRA, giving 0.93% (95% CI 0.93±0.001) as the percentage of patients presenting to the ED caused by isolated ADRs. The mean age of patients with ADRs was 48.85 years and mostly were Type A. Females accounted for the majority of ADRs cases, representing 61.8% (134 patients) of the cohort compared to 38.2% for males. The risk of ADRs is relatively evenly distributed across middle-to-older age groups, with the 45–54 and 55–64 age brackets each accounting for 27.2% of cases. Using the Hartwig’s criteria, it was seen that 152/217 (70%) patients had mild-moderate ADRs while 65/217 (30%) had severe ADRs. Thus, a majority of the ADRs detected were of mild-moderate severity (Table 1). It was seen that 144 (66.4%) events were type A ADRs which possibly avoidable while 73 (33.6%) were Type B ADRs which possible unavoidable. Demographics data was shown in Table 1.
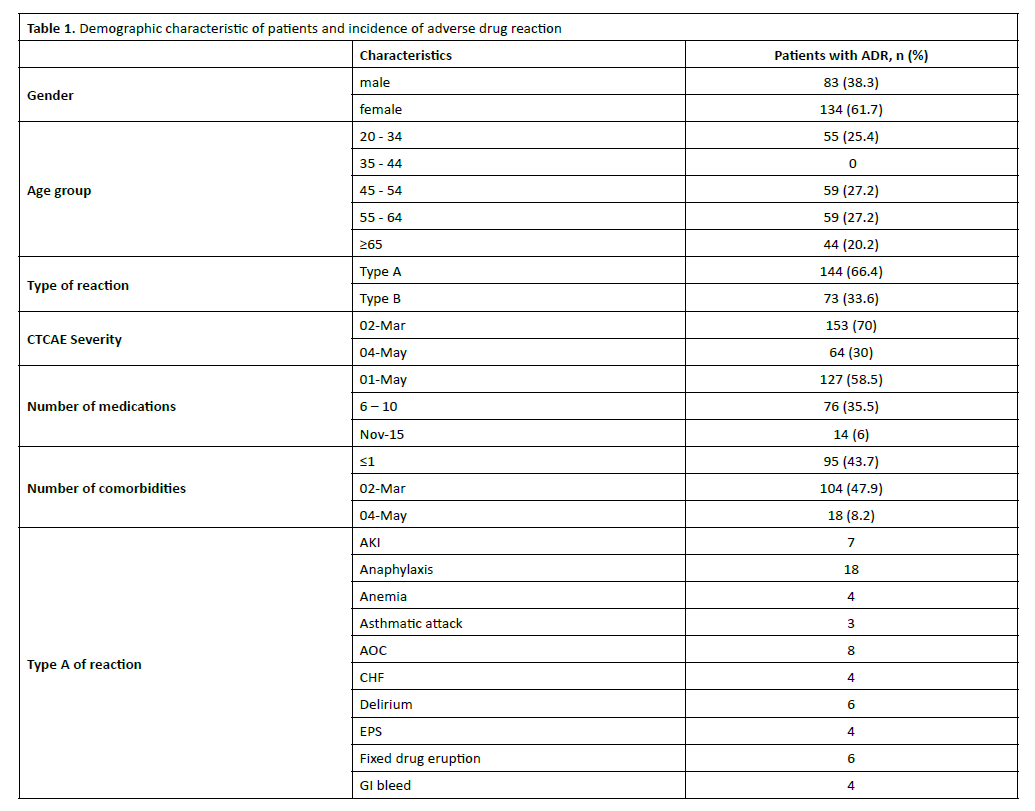
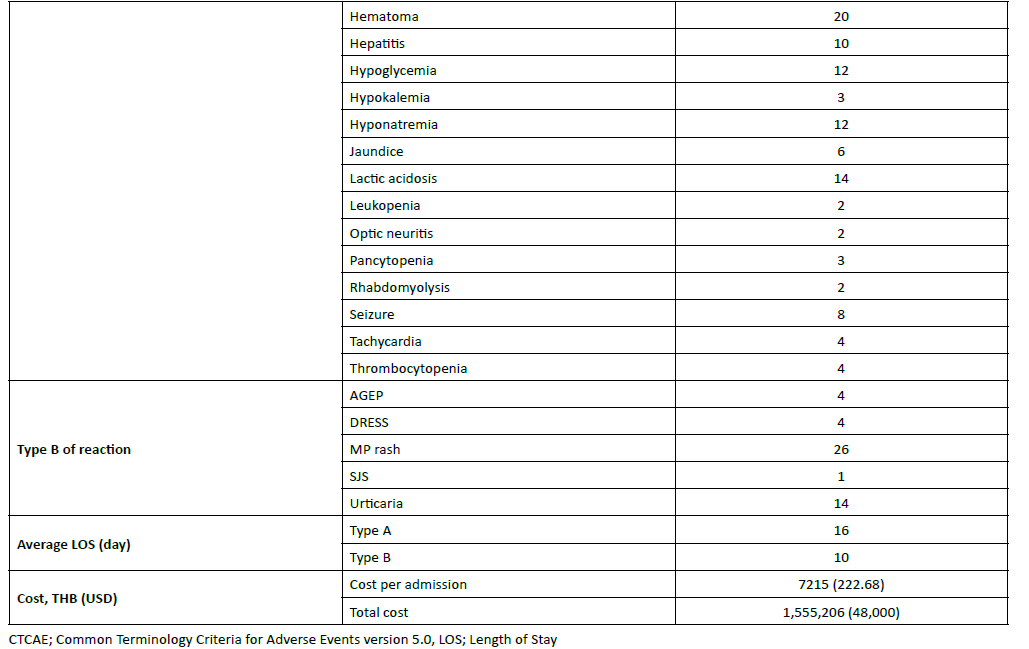
Our study distinguishes between two primary types of reactions: Type A ADRs, mostly preventable ADRs, predictable and often dose-dependent reactions, were the most common, occurring in 66.4% of patients. Examples from the table include acute kidney injury (7 cases) and gastrointestinal bleeding (4 cases). While Type B, mostly unpredictable and idiosyncratic reactions (often immune-mediated) occurred in 33.6% of patients. Notable examples include anaphylaxis (18 cases) and fixed drug eruption (6 cases). Interestingly, the average length of stay (LOS) varied significantly by reaction type. Patients experiencing Type A ADRs had a mean stay of 5 days, whereas those with Type B reactions required a substantially longer hospitalization of 16 days. The majority of reported reactions severity (70%) were categorized as Grade 2–3, which typically signifies mild to moderate reactions requiring medical intervention or hospitalization. The remaining 30% were severe (Grade 4–5).
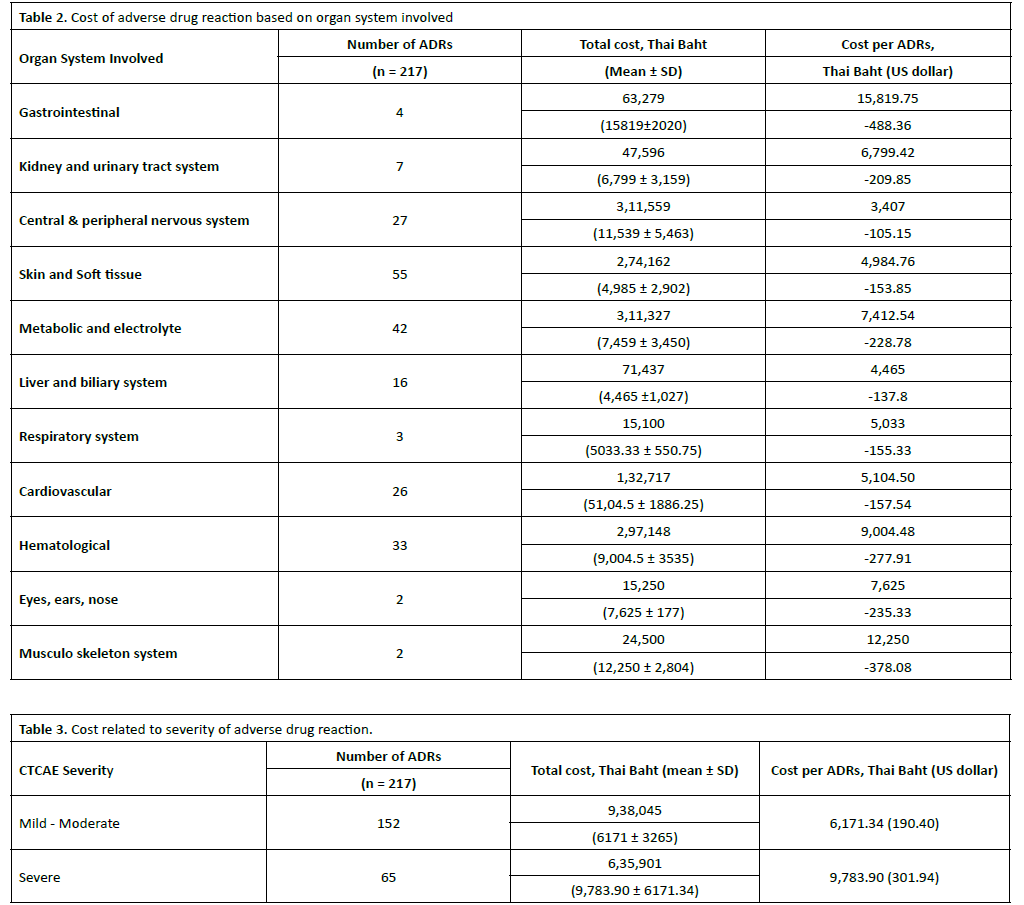
The common medication classes involved in ADRA included antimicrobial agent (59/217, 27.18%), drugs used in diabetes (31/217, 14.28%), non-steroidal anti-inflammatory drugs (29/217, 13.36%), Anti-thrombotic agents (20/217, 9.21%), Anti-psychotic drug (19/217, 8.75%), Anti-convulsant (16/217, 7.37%), while out of 217 events, 144 (66.35%) were identified as the potential preventability of ADRA. The mean hospital stay of patients with ADRA was 15 days. Average length of stay for Type A and Type B ADRs was 16 and 10 days respectively. The total cost for 217 patients with ADRA was 1,555,206 THB (48,000 USD). The mean total cost per patient with ADRA was estimated at 7215 THB (222.68 USD) (Table 2-Table 3).
The data provided in Table 2 outlines the economic burden of ADRs across various organ systems, measured in Thai Baht (THB) and US Dollars (USD). Based on the 217 recorded ADRs, the skin and soft tissue system recorded the highest number of cases (55 cases), followed by metabolic and electrolyte issues (42 cases) and hematological issues (33 cases). The highest total costs, despite being second in frequency, the central and peripheral nervous system incurred the highest total cost at 311,559 THB. This is closely followed by metabolic and electrolyte (311,327 THB) and hematological systems (297,148 THB). Focusing on the cost per individual ADRs based on organ systems, the gastrointestinal system is the most expensive to manage on a per-case basis (488.36 USD), costing more than triple the amount of a skin and soft tissue ADRs (153.85 USD), even though gastrointestinal cases were relatively rare (only 4 cases). Interestingly, while the central and peripheral nervous system had a high total cost, its cost per individual ADRs was the lowest of all systems listed at 3,407 THB (105.15 USD). This suggests that while these reactions are common or aggregate to a high cost, the individual management of each case is relatively less resource-intensive. Our data suggests that, the metabolic and hematological systems represent a significant portion of the total economic burden due to the high volume of reported ADRs and the escalating clinical expenditures within the hospital. Nevertheless, gastrointestinal and musculoskeletal systems are rare but represent the most significant financial hit per occurrence. On the contrary, skin and nervous system ADRs are frequent but are among the least expensive to treat individually. Our data suggests a tiered burden, necessitating targeted interventions for high-volume, high-cost ADRs events to optimize hospital resource allocation and minimize preventable expenditures.
The total economic burden for managing both Type A and Type B adverse drug reactions was 1,564,075 THB (48,273 USD). The mean cost per admission was calculated at 7,207.71 THB (222.46 USD). The costs associated with ADRs, categorized by severity (Table 3), outlines the economic impact of 217 recorded ADRs cases, categorized by the Common Terminology Criteria for Adverse Events (CTCAE). The mild ADRs are significantly more common, accounting for 152 cases, while the moderate ADRs occurred 65 cases. For total expenditure, the total cost for mild ADRs reached 938,045 THB, whereas moderate ADRs totaled 635,901 THB. Remarkably, the mean cost per case for a mild – moderate ADRs is 6,171.34 THB (190.40 USD), while a severe ADRs is notably higher at 9,783.90 THB (301.94 USD). These figures reveal a clear correlation between the severity of the reaction and the financial burden per incident.
DISCUSSION
In a hospital setting, physician serves as the primary clinical decision-maker and the first line of defense in an ADRs prevention system. While pharmacists manage the logistics and nurses monitor the administration, doctors are responsible for the diagnostic, because ADRs often mimic disease states, physician are uniquely qualified to differentiate between a worsening illness and a drug-induced event. Pharmacist, as the co-leader, roles in a modern hospital system, the prevention and detection of ADRs is a complex, multi-layered system that
combines human expertise with advanced digital surveillance. Pharmacists serve as the primary safeguards against ADRs. Their role spans the entire medication-use process, from the initial admission to post-discharge follow-up. By integrating clinical expertise with real-time patient data, including medication reconciliation on admission, order verification in every prescription for drug-drug/drug-food interactions, highalert drugs which have a high risk of causing significant harm if misused, detection involves active surveillance and trigger monitoring rather than waiting for an event to be reported. Once pharmacist together with physician shared decision for ADRs diagnosis, using the Naranjo’s algorithm to determine the likelihood of the drug being the cause. The severity of ADRs and its root cause were added to the patient’s profile. This data helps identify “signals” for new, previously unknown side effects to prevent future re-exposure.
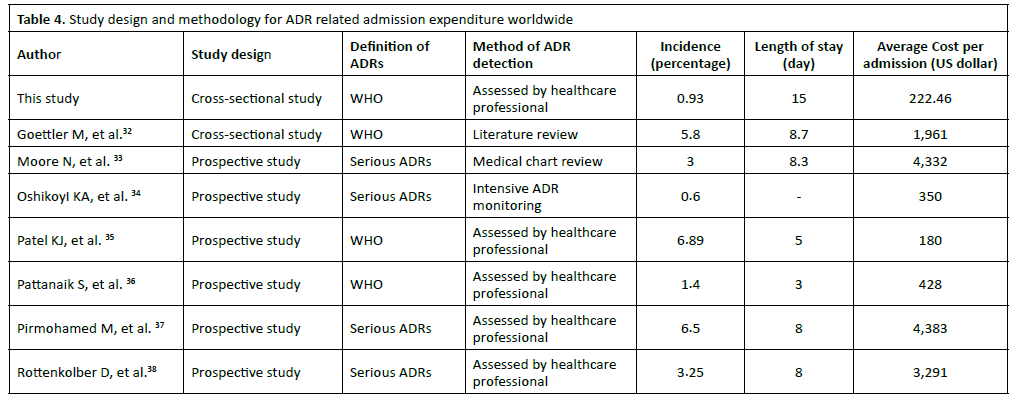
In light of the increasing complexity of pharmacotherapy and an ageing population exposed to the risk of multimorbidity and polypharmacy, ADRs remain a major public health issue with a significant impact on healthcare expenditure, but it could be avoided and potential cost savings were very wide26- 31. The findings of this study provide critical insights into the clinical and economic impact of adverse drug reactions (ADRs) within the local context, particularly when compared to global benchmarks32-38. Based on the data provided in the comparative table (Table 4), comparing our findings regarding incidence, length of stay (LOS), and economic burden with global literature. The incidence and economic impact of adverse drug reactions (ADRs) found in this study vary significantly when compared to international data, likely reflecting differences in study settings, patient populations, and ADR definitions. The ADRs incidence in this study was 0.93%, which is relatively low compared to several prospective studies. Osanlou R, et al.37 reported an incidence of 6.5%. Lagnaoui R, et al.30 and Patel KJ, et al.35 observed rates of 7% and 6.89%, respectively. Conversely, our incidence is more closely aligned with, though slightly higher than, the 0.6% reported by Oshikoya KA, et al.34. The lower incidence observed in our study compared to Osanlou R, et al. or Lagnaoui R, et al. may be due to the method of detection used here, which might capture fewer events than the intensive monitoring or systematic medical chart reviews used in another research.
A striking finding in our study is the average length of stay (LOS) of 15 days, which is considerably longer than all other studies listed in the comparison. Most prospective studies, such as those by Moore N, et al.33 (8.3 days) and Rottenkolber D, et al.38 (8 days), reported LOS values roughly half as long as ours. The shortest LOS was noted by Pattanaik S, et al.36 at 3 days. The extended 15-day stay in our population suggests that while the incidence is low, the complications arising from these ADRs are severe or occur in patients with complex underlying conditions that require prolonged stabilization. For economic perspective, the average cost per ADR admission in this study was 222.46 USD. This demonstrates a clear geographic and systemic divide in healthcare spending. Our costs are similar to country in developing regions, in India (180 USD, reported by Patel KJ, et al.)35 and Nigeria (350 USD, reported by Oshikoya KA, et al.)34. There is a vast difference when compared to European data, where Moore N, et al.33 and Osanlou R, et al.37 reported costs exceeding 3,291 - 4,300 USD per admission. While our percase cost is relatively low in absolute US dollars, the burden is magnified by the significantly longer hospital stay (15 days), which consumes institutional resources such as bed occupancy and nursing care. The combination of a lower per-admission cost with a substantially higher LOS suggests that ADRs in our setting may lead to inefficiencies in hospital bed utilization and prolonged patient recovery times.
Strengths and Limitations
Our study provides additional features to strengthen study design, ADRs screening in all those admitted to the hospital caused by ADRs with the appropriate sample size for logically survey research. Including the elaborate and careful identification of an ADRs on the first few days of admission by ADR specialized pharmacist together with physician. However, there are a number of limitations. there were differences in key disease areas, co-existing conditions in an individual, which may also have had an effect on the utilization of health care resources and healthcare expenditure. In addition, the comprehensive collection of healthcare resource use before and after hospital admission was not included. Comparisons with other studies are difficult due to differences in cost sources. However, that ADRs survey was the most comprehensive to date in terms of patient characteristics. The causality assessment of ADRs is subjective. However, we have reduced this by having two independent assessors, pharmacist specialized in ADR, and physician to resolve any discrepancies. Our data was from a single institution and the types of patients in our setting could be different from that in other hospitals, leading to the underor over-estimated of cost burden. Therefore, generalizability to other populations and settings may be limited, related to estimates of costs which are based on estimated time and units costs available. The costs associated with ADRA modest, but also depend on the severity of the ADRs. However, the cost is not only financial but is often associated with costs both clinically and personally for individuals. Early intervention, where possible, is important to avoid preventable medication harm. Intervention with more targeted policies to reduce ADRs through identification of those at highest risk and more awareness among populations at risk will help to reduce costly hospitalizations and avoid increased morbidity and mortality.
CONCLUSION
In conclusion, ADRA results in significant healthcare utilization and substantial economic burdens. The costs are increased with preventability and severity of the ADRs, drawing attention of hospital administration and health care policy makers to save the unnecessary cost. More research is required to develop validated tools for prevention and early detection of ADRs that can be implemented in clinical practice to avoid unnecessary harm and economic burden.
AUTHOR CONTRIBUTIONS
Conceptualization: Monton Tanabodee
Data Curation: Monton Tanabodee, Nirun Jangkong
Formal Analysis, Investigation and Methodology: Monton Tanabodee, Nirun Jangkong
Project Administration and Resources: Monton Tanabodee
Supervision: Monton Tanabodee
Writing: Monton Tanabodee, Nirun Jangkong
The final form of the manuscript: All authors.
COMPETING INTERESTS
All authors have no competing interests
References
- Khangtragool A, Nukompun K, Teeyasuntranon A, Wannasiri P, Moraray S, Khangtragool W. Evaluation of rational drug use based on World Health Organization indicators in a tertiary hospital, Thailand. Pharm Sci Asia. 2023;50(1):1-8.
- Riedl MA, Casillas AM. Adverse drug reactions: types and treatment options. Am Fam Physician. 2003;68(9):1781-1791.
- Harmark L, van Grootheest AC. Pharmacovigilance: methods, recent developments and future perspectives. Eur J Clin Pharmacol. 2008;64(8):743-752.
- World Health Organization. The importance of pharmacovigilance. Geneva: World Health Organization; 2002.
- World Health Organization. WHO pharmacovigilance indicators: a practical manual for the assessment of pharmacovigilance systems. Geneva: World Health Organization; 2015.
- Suke SG, Kosta P, Negi H. Role of pharmacovigilance in India: an overview. Online J Public Health Inform. 2015;7(2):e223.
- Sawanpanyalert P, Suwankesawong W. Health products vigilance in Thailand: past, present and future. J Health Sci Thai. 2024;25(3):444-455.
- Li R, Curtain C, Bereznicki L, Zaidi STR. Community pharmacists’ knowledge and perspectives of reporting adverse drug reactions in Australia: a cross-sectional survey. Int J Clin Pharm. 2018;40(4):878-889.
- Yu YM, Shin WG, Lee JY, Choi SA, Jo YH, Youn SJ, et al. Patterns of adverse drug reactions in different age groups: analysis of spontaneous reports by community pharmacists. PLoS One. 2015;10(7):e0132916.
- Thai Food and Drug Administration, Health Product Vigilance Center. Statistic of adverse events during 1984–2019.
- Akaleephan C, Kaewpanukrunsi W, Limwattananon C. Adverse drug reaction monitoring program. J Health Sci. 2004;13(2):350-361.
- Chiewchantanakit D. Study of adverse drug reactions at Queen Sirikit National Institute of Child Health [dissertation]. Bangkok: Mahidol University; 2000.
- Siltharm C, Pattanaprateep O, Pongcharoensuk P, Jeanpeerapong N, Thavorncharoensap M. Detection of adverse drug reaction (ADR)-related hospital admissions: a pilot study using administrative database for ADR monitoring in Thailand. Pharm Sci Asia. 2017;44(3):142-153.
- Tragulpiankit P, Pummangura C, Luscombe DK, Kaojarern S, Wananukul W, Montakantikul P, et al. Prevention of adverse drug reactions in hospitalised patients by pharmacist participation at a large teaching hospital in Thailand. Drug Saf. 2005;28(10):931-944.
- Sittiphan S, Lim A, Khurram H, Dureh N, Dittakan K. Epidemiology of reported serious adverse drug reactions due to anti-infectives using nationwide database of Thailand. PLoS One. 2025;20(2):e0318597.
- Kampichit S, Srisuriyachanchai W, Pratipanawatr T, Jarernsiripornkul N. Accuracy in patient-reported adverse drug reactions and their recognition: a mixed-methods study. Int J Clin Pharm. 2024;46(2):401-410.
- Edwards IR, Aronson JK. Adverse drug reactions: definitions, diagnosis, and management. Lancet. 2000;356(9237):1255-1259.
- Rajakannan T, Mallayasamy S, Rajesh V, Muralidhar V, Smitha P, Sudha V. Development of indicators for identifying adverse drug events in an Indian tertiary care teaching hospital. Drug Healthc Patient Saf. 2010;2:95-100.
- Naranjo CA, Busto U, Sellers EM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30(2):239-245.
- Rawlins MD, Thompson JW. Pathogenesis of adverse drug reactions. In: Davies DM, editor. Textbook of adverse drug reactions. Oxford: Oxford University Press; 1977. p. 10.
- Hartwig SC, Siegel J, Schneider PJ. Preventability and severity assessment in reporting adverse drug reactions. Am J Hosp Pharm. 1992;49(9):2229-2232.
- WHO Collaborating Centre for Drug Statistics Methodology. ATC classification system. Oslo: WHO Collaborating Centre for Drug Statistics Methodology.
- Uppsala Monitoring Centre. WHO adverse reaction terminology. Uppsala: Uppsala Monitoring Centre; 2007.
- Ngamjarus C, Chongsuvivatwong V, McNeil E. n4Studies: sample size calculation for an epidemiological study on a smart device. Siriraj Med J. 2016;68(3):160-170.
- Thai Food and Drug Administration, Health Product Vigilance Center. Spontaneous reports of adverse drug reactions 2022.
- Batel Marques F, Penedones A, Mendes D, Alves C. A systematic review of observational studies evaluating costs of adverse drug reactions. Clinicoecon Outcomes Res. 2016;8:413-426.
- Formica D, Sultana J, Cutroneo PM, Lucchesi S, Angelica R, Crisafulli S, et al. The economic burden of preventable adverse drug reactions: a systematic review of observational studies. Expert Opin Drug Saf. 2018;17(7):681-695.
- Gautier S, Bachelet H, Bordet R, Caron J. The cost of adverse drug reactions. Expert Opin Pharmacother. 2003;4(3):319-326.
- Bordet R, Gautier S, Le Louet H, Dupuis B, Caron J. Analysis of the direct cost of adverse drug reactions in hospitalized patients. Eur J Clin Pharmacol. 2001;56(12):935-941.
- Lagnaoui R, Moore N, Fach J, Longy-Boursier M, Bégaud B. Adverse drug reactions in a department of systemic diseases-oriented internal medicine: prevalence, incidence, direct costs and avoidability. Eur J Clin Pharmacol. 2000;56(2):181-186.
- Jolivot PA, Pichereau C, Hindlet P, Hejblum G, Bigé N, Maury E, et al. An observational study of adult admissions to a medical ICU due to adverse drug events. Ann Intensive Care. 2016;6(1):9.
- Goettler M, Schneeweiss S, Hasford J. Adverse drug reaction monitoring cost and benefit considerations part II: cost and preventability of adverse drug reactions leading to hospital admission. Pharmacoepidemiol Drug Saf. 1997;6 Suppl 3:S79-S90.
- Moore N, Lecointre D, Noblet C, Mabille M. Frequency and cost of serious adverse drug reactions in a department of general medicine. Br J Clin Pharmacol. 1998;45(3):301-308.
- Oshikoya KA, Chukwura H, Njokanma OF, Senbanjo IO, Ojo I. Incidence and cost estimate of treating pediatric adverse drug reactions in Lagos, Nigeria. Sao Paulo Med J. 2011;129(5):317-323.
- Patel KJ, Kedia MS, Bajpai D, Mehta SS, Kshirsagar NA, Gogtay NJ. Evaluation of the prevalence and economic burden of adverse drug reactions presenting to the medical emergency department of a tertiary referral center: a prospective study. BMC Clin Pharmacol. 2007;7:8.
- Pattanaik S, Dhamija P, Malhotra S, Sharma N, Pandhi P. Evaluation of cost of treatment of drug-related events in a tertiary care public sector hospital in Northern India: a prospective study. Br J Clin Pharmacol. 2009;67(3):363-369.
- Osanlou R, Walker L, Hughes DA, Burnside G, Pirmohamed M. Adverse drug reactions, multimorbidity and polypharmacy: a prospective analysis of 1 month of medical admissions. BMJ Open. 2022;12(7):e055551.
- Rottenkolber D, Schmiedl S, Rottenkolber M, Farker K, Saljé K, Mueller S, et al. Adverse drug reactions in Germany: direct costs of internal medicine hospitalizations. Pharmacoepidemiol Drug Saf. 2011;20(6):626-634.
