Possible Effect of Allopurinol and Risk of Dementia: An Updated Meta-Analysis
Ahmed Alturqi Alenezi. Department of Clinical Pharmacology, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia. aalenezi0046@stu.kau.edu.sa
Mai A. Alim A. Sattar Ahmad. Department of Clinical Pharmacology, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia. maiahmad@kau.edu.sa
Ibrahim M. Ibrahim. Department of Clinical Pharmacology, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia. imibrahim1@kau.edu.sa
DOI: 10.18549/PharmPract.2026.1.3416
Abstract
Background: Allopurinol is widely prescribed for the management of gout and hyperuricemia through inhibition of xanthine oxidase. While uric acid possesses antioxidant properties, xanthine oxidase activity contributes to oxidative stress, raising uncertainty regarding the long-term cognitive effects of allopurinol therapy. The association between chronic allopurinol use and dementia risk remains unclear. This updated meta-analysis aimed to evaluate the association between long-term allopurinol exposure and the risk of incident dementia in patients with gout or hyperuricemia. Methods: This systematic review and meta-analysis was conducted and reported in line with PRISMA reporting principles. PubMed, MEDLINE, and the Cochrane Library were systematically searched for observational studies published between January 2015 and August 2024. Study quality was assessed using the Newcastle–Ottawa Scale. Adjusted hazard ratios (HRs) derived from time-to-event analyses were prespecified as the primary effect measure, reflecting longitudinal dementia risk, while adjusted odds ratios (ORs) were analyzed separately as a secondary outcome. Meta-analyses were performed using a random-effects model in Review Manager (RevMan) version 5.4. Results: Nine studies met the inclusion criteria, comprising cohort, case-control, and nested case-control designs. In the primary HR-based meta-analysis, long-term allopurinol use was not associated with an increased risk of dementia, with substantial heterogeneity observed. Sensitivity analyses excluding one influential cohort study demonstrated a statistically significant reduction in incident dementia, with reduced heterogeneity. Across individual studies, most effect estimates were below unity, suggesting a neutral-to-potentially protective association. OR-based analyses were limited in number and showed no consistent association. Conclusion: Current evidence indicates that long-term allopurinol therapy is associated with a reduced incidence of dementia in populations, as suggested by sensitivity analyses. These findings support the cognitive safety of allopurinol in routine clinical practice and demonstrate the value of pharmacist-led medication review and longitudinal monitoring. Further well-designed prospective studies are warranted to clarify causality and identify subgroups most likely to benefit.
Keywords: allopurinol, gout, dementia, xanthine oxidase, uric acid, and cognitive function
INTRODUCTION
Gout is a metabolic disorder characterized by elevated serum uric acid (SUA) levels and is increasingly associated with cardiovascular morbidity and cognitive impairment1. SUA has antioxidant properties that could theoretically protect the brain2,3, but its link to dementia is still up for debate. Epidemiological studies have reported conflicting findings, with some suggesting that higher SUA levels are associated with a lower risk of dementia3,4, while others link hyperuricemia to accelerated cognitive decline and neurodegeneration5,6. These inconsistencies emphasize the need to clarify whether dementia risk is driven by uric acid itself or by its pharmacological modulation.
Allopurinol, a xanthine oxidase inhibitor widely prescribed for gout and hyperuricemia, effectively lowers SUA concentrations7. However, its long-term cognitive effects are not well defined. From a mechanistic perspective, allopurinol may influence dementia risk through competing pathways: on one hand, lowering SUA could attenuate endogenous antioxidant defenses; on the other, inhibition of xanthine oxidase may reduce oxidative stress and neuroinflammatory signaling implicated in neurodegenerative processes8,9. Clinical evidence remains inconsistent, with some observational studies reporting increased dementia risk among long-term allopurinol users10, while others describe neutral or potentially protective associations11,12. Resolving these discrepancies is essential for informed clinical decision-making.
Dementia represents a growing global health challenge. Global Burden of Disease estimates suggest that approximately 55 million people are living with dementia, and prevalence is projected to rise substantially by 205013. Alzheimer’s disease and vascular dementia, the most common subtypes, arise from complex interactions involving oxidative stress, metabolic dysregulation, vascular pathology, and genetic susceptibility14. Identification of modifiable risk factors, including commonly prescribed medications such as allopurinol, is therefore of major public health relevance.
Despite the established efficacy of allopurinol in gout management15, its dual biological effects complicate long-term risk–benefit assessment. While SUA reduction may theoretically weaken antioxidant capacity, xanthine oxidase inhibition may mitigate oxidative injury and vascular dysfunction16. Existing meta-analyses addressing urate-lowering therapy and cognitive outcomes are limited by narrow scope, small sample sizes, heterogeneous outcome definitions, and incomplete adjustment for confounders such as age, comorbidity burden, treatment duration, and renal function17-21. Moreover, several large population-based studies published in recent years have not been comprehensively synthesized, leaving uncertainty regarding the cognitive safety of long-term allopurinol therapy.
Relevance to pharmacy practice: In routine clinical care, allopurinol is frequently initiated and continued for prolonged periods, particularly among older adults with multimorbidity, including chronic kidney disease (CKD), cardiovascular disease, diabetes, and polypharmacy. Pharmacists play a central role in comprehensive medication reviews (CMRs) and medication therapy management (MTM), supporting adherence, titrating doses toward serum urate targets, monitoring safety, and coordinating dose adjustments with prescribers. Clarifying whether long-term allopurinol exposure is associated with incident dementia therefore has direct implications for pharmacy practice, including cognitive monitoring during follow-up, informed risk–benefit discussions, and dose optimization in vulnerable populations.
This study is framed as a pharmacoepidemiologic drug-safety meta-analysis evaluating unintended cognitive outcomes associated with chronic therapy. We systematically examined the association between long-term allopurinol use and incident dementia, prioritizing dementia onset as a time-toevent outcome. Accordingly, adjusted hazard ratios (HRs) were prespecified as the primary effect measure, as they capture both the occurrence and timing of dementia and are particularly relevant for pharmacists involved in longitudinal medication monitoring. Secondary analyses based on odds ratios (ORs) were considered separately when time-to-event estimates were unavailable.
The objectives of this meta-analysis were to:
- Assess whether allopurinol use is associated with increased or decreased dementia incidence among patients with gout or hyperuricemia.
- Explore potential mechanisms distinguishing effects related to SUA reduction from direct pharmacological actions of xanthine oxidase inhibition.
- Identify subgroups that may experience differential effects, including older adults, patients with higher doses or longer treatment duration, and region-specific populations.
By synthesizing contemporary observational evidence, this study aims to clarify the cognitive safety profile of long-term allopurinol therapy and to inform medication management strategies for patients with gout who are at risk of dementia.
METHODS
This systematic review and meta-analysis was conducted and reported in line with PRISMA reporting principles. As the analysis used previously published, de-identified data, additional ethical approval was not required. Observational designs (cohort, case-control, and nested case-control) were eligible because they address the same clinical question in comparable populations; however, effect measures were not pooled across designs when they were not comparable. Specifically, adjusted hazard ratios (HRs) from time-to-event models were prespecified for the primary meta-analysis, while adjusted odds ratios (ORs) from case-control or nested case-control studies were analyzed separately to avoid effectmeasure mismatch. Where heterogeneity was expected, random-effects models were applied, and prespecified subgroup and sensitivity analyses were used to explore sources of variability22.
PICO Framework
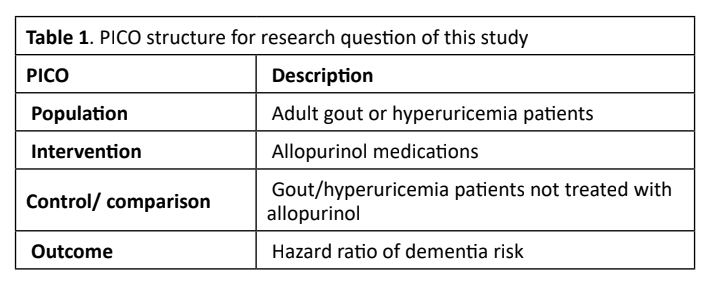
What are the long-term implications of allopurinol treatment on dementia risk in patients with gout or hyperuricemia? This research employed the Population Intervention Control Outcome (PICO) framework to direct the investigation (Table 1)23.
Search strategy
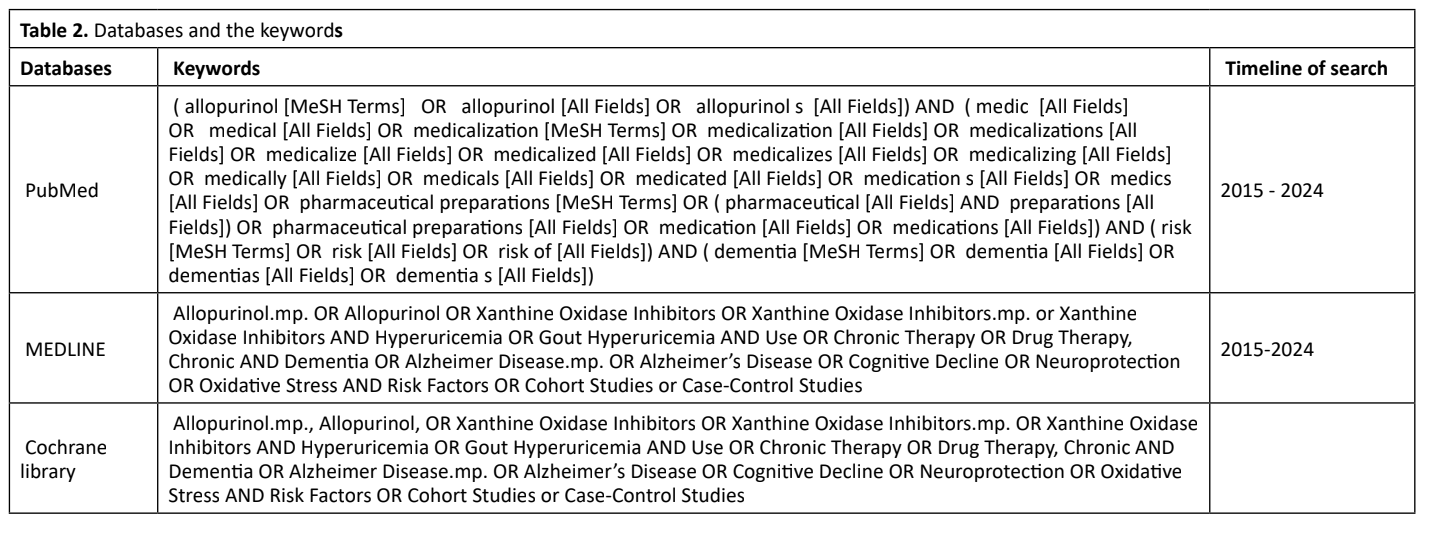
A systematic search was conducted in PubMed/MEDLINE and the Cochrane Library for eligible observational studies published from 1 January 2015 to 31 August 2024. The strategy combined controlled vocabulary and free-text terms for the exposure and outcome, including allopurinol, xanthine oxidase inhibitor, gout, hyperuricemia, dementia, Alzheimer disease, and related terms. Search strings were adapted to each database and are provided in Table 2. Reference lists of included articles and relevant reviews were hand-searched to identify additional eligible studies. Records were deduplicated prior to screening. Two reviewers independently screened titles/abstracts and then full texts; disagreements were resolved by discussion.
Eligibility Criteria
Inclusion Criteria:
Studies were eligible if they:
- included adults with gout or hyperuricemia;
- evaluated allopurinol exposure (e.g., allopurinol users vs non-users, or allopurinol vs other urate-lowering therapy) or reported extractable data relevant to long-term urate lowering therapy in gout/hyperuricemia populations
- reported incident dementia (all-cause dementia and/or Alzheimer’s disease) as an outcome;
- used an observational design (cohort, case-control, or nested case-control);
- provided adjusted effect estimates (HR or OR) with 95% CIs or sufficient data to derive them;
- were published in English with full text available.
In addition to studies directly evaluating allopurinol exposure, we also included large population-based cohort studies in which gout or hyperuricemia served as the primary exposure. These studies were retained because gout represents the principal clinical indication for long-term allopurinol therapy, and such cohorts inherently comprise a high proportion of individuals exposed to urate-lowering treatment. Inclusion of these studies allowed contextual interpretation of dementia risk within populations for whom allopurinol is routinely prescribed, while acknowledging that the reported effect estimates reflect disease exposure rather than medication exposure per se. These studies were not considered direct estimates of the effect of allopurinol itself but rather as complementary evidence describing dementia risk in populations in which chronic allopurinol use is common.
Studies were categorized into two groups: (1) studies directly comparing allopurinol exposure versus non-exposure; and (2) studies where gout or hyperuricemia was the primary exposure (indication-based cohorts). Because the second group does not estimate the medication effect directly, these studies were used for contextual interpretation and were not treated as direct evidence of allopurinol’s causal effect.
Exclusion Criteria
Studies were excluded if they met any of the following criteria.
- Focused on populations other than patients with gout or hyperuricemia.
- Assessed clinical outcomes unrelated to dementia risk.
- They were categorized as systematic reviews, metaanalyses, literature reviews, observational studies without longitudinal data, scoping reviews, conference abstracts, or editor letters.
- Published in languages other than English (e.g., Chinese, Arabic, Spanish, or German).
- Duplicate publications or studies with no full-text availability.
Rationale for Selection
The inclusion and exclusion criteria were designed to ensure that only studies directly addressing the core research question.
Whether allopurinol use in gout patients is associated with dementia risk was considered. This approach also aimed to minimize bias by excluding irrelevant or low-quality studies.
Data Extraction
Following the screening process, two independent researchers selected studies for inclusion. Any discrepancies were addressed via dialogue or with the participation of an additional evaluator. The data obtained from each trial are the following:
- Authors and year of publication.
- Country where the study was conducted
- Study population and sample size. • Follow-up duration.
- A dose of allopurinol was administered.
- Reported hazard ratios (HR) or odds ratios (OR) for dementia risk.
- Exposure definition (how each study defined “allopurinol use”, duration, dose, adherence)
- Outcome definition (ICD codes / registries / validated diagnosis)
This information was systematically organized and prepared for pooled analysis to ensure consistency and transparency.
Outcomes and effect measures
The primary outcome was incident dementia, summarized as adjusted hazard ratios (HRs) with 95% confidence intervals (CIs). HRs were prespecified as the primary effect measure because dementia is a time-to-event outcome, and HRs account for variable follow-up duration and censoring across cohorts. This is clinically relevant for pharmacy practice, where patients receiving urate-lowering therapy are monitored longitudinally and exposure may change over time. As a secondary analysis, adjusted odds ratios (ORs) were extracted only when HRs were not reported. ORs were pooled separately to avoid mixing effect measures and to maintain clinical interpretability.
Brief reports or letters that reported effect estimates but lacked sufficient methodological detail for pooling were tabulated for completeness but excluded from quantitative meta-analysis.
Target Population:
- Treatment Group: Gout patients treated with allopurinol.
- Control Group: Gout patients not receiving allopurinol.
1.Outcome Assessment:
- New diagnoses of dementia.
- Time to dementia onset.
- Safety-related outcomes associated with allopurinol use.
2.Factors Influencing Outcomes:
- Duration of allopurinol use.
- Dosage of allopurinol.
- Patient characteristics and demographics (e.g., age, gender, and comorbidities).
These outcomes were selected to comprehensively evaluate the likely connection between allopurinol use and dementia risk while accounting for important influencing factors.
Quality Assessment
Two reviewers independently evaluated research quality utilizing the Newcastle-Ottawa Scale (NOS), which is wellestablished for assessing the quality of non-randomized investigations. The Newcastle-Ottawa Scale (NOS) is a standardized instrument for evaluating the quality of nonrandomized studies, including cohort and case-control studies. It evaluates studies based on three critical areas: how participants are selected, how groups are compared to control for confounding factors, and how outcomes are measured and followed up. By providing a structured approach to assessing study quality, the NOS helps ensure that solely methodologically sound and reliable studies are included in systematic reviews and meta-analyses.
(The NOS focuses on three key domains:
- Selection: The representativeness of the exposed group, the selection of the unexposed group, ascertainment of exposure, and confirmation that the outcome of interest
- Comparability: Control for confounding factors, including both primary and additional factors.
- Outcome: The assessment of outcomes, adequacy of follow-up duration, and completeness of follow-up data.
Each study was scored on a scale of 0 to 9, with higher scores indicating higher quality. The classification system used was as follows:
- Studies with a score of 7 or higher were considered low risk of bias.
- Studies scoring between 5 and 7 were classified as moderate risk of bias.
- Studies with scores <5 were considered high risk and were retained for sensitivity/subgroup analyses.
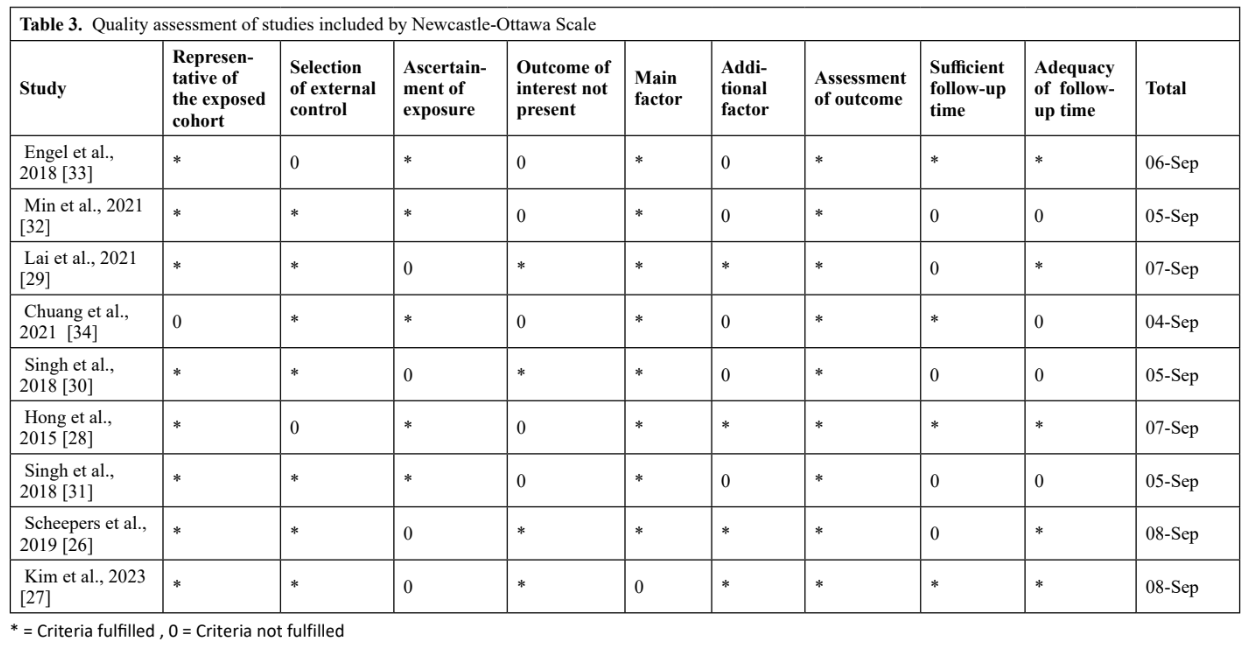
Disagreements between reviewers regarding quality assessment were resolved through discussion, and a consensus was reached. Table 3 summarizes the quality assessment results.
Statistical analysis
Statistical analyses were performed using Review Manager (RevMan), version 5.4. The primary meta-analysis pooled adjusted hazard ratios (HRs) with corresponding 95% confidence intervals (CIs), as HRs are the most appropriate effect measure for time-to-event outcomes such as incident dementia and account for differences in follow-up duration and censoring across cohorts. Only studies reporting HRs derived from longitudinal survival analyses (e.g., Cox proportional hazards models) were included in the primary synthesis.
Studies reporting adjusted odds ratios (ORs) from logistic or conditional logistic regression models were not combined with HRs and were analyzed separately in a secondary OR-based meta-analysis in order to avoid mixing non-comparable effect measures and to preserve clinical interpretability.
Between-study heterogeneity was assessed using Cochran’s Q test and the I² statistic. A random-effects model was applied when heterogeneity was substantial (I² > 50% or Q-test p < 0.10); otherwise, a fixed-effect model was used. Prespecified sensitivity analyses were conducted to evaluate the influence of individual studies with disproportionate statistical weight, including leave-one-out analyses to assess the robustness of the pooled estimates.
Assessment of Publication Bias
Assessment of publication bias was restricted to the primary HR-based meta-analysis, as HRs were prespecified as the principal effect measure for the primary outcome. Funnel plots were constructed by plotting the log-transformed HRs against their standard errors (SEs), and asymmetry was evaluated by visual inspection.
Formal statistical tests for funnel plot asymmetry (e.g., Egger’s tests) were not performed, because fewer than 10 studies were
included in the secondary analysis.
Quality Assessment
As shown in Table 3 and Table 4, among the nine included studies, four studies were of low risk26-29, four studies were of moderate risk30-33, and one study was of high risk34.
Subgroup and Sensitivity Analyses (HR-based studies)
All subgroup and sensitivity analyses described below were restricted to studies reporting adjusted hazard ratios (HRs) derived from longitudinal time-to-event models. Studies reporting odds ratios (ORs) from case-control or nested casecontrol designs were not pooled with HRs and were instead addressed in the secondary analysis or described narratively in order to avoid effect-measure mismatch.
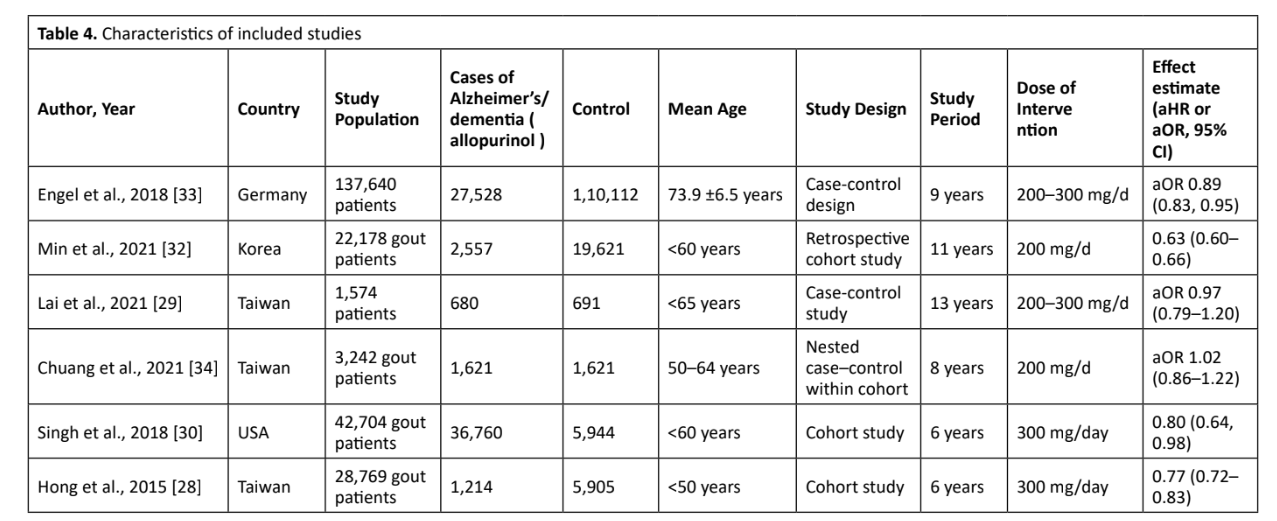
Study Quality (Newcastle–Ottawa Scale): When analyses were restricted to high-quality cohort studies (NOS ≥ 7; n = 3 HR studies: Scheepers 2019; Hong 2015; Kim 2023), a consistent protective association between allopurinol use and incident dementia was observed, with moderate heterogeneity. These studies contributed substantially to the stability of the pooled estimate.
In contrast, moderate-quality studies (NOS 5–6; n = 3 HR studies: Min 2021; Singh 201830; Singh 201831) demonstrated substantial heterogeneity, largely driven by one very large U.S. cohort that reported an increased dementia risk. When this influential study was excluded in prespecified sensitivity analyses, the pooled estimate shifted toward a statistically significant protective association, and heterogeneity decreased markedly. No low-quality HR-based studies (NOS < 5) contributed to the primary pooled analysis.
Study Design: All estimates included in the primary metaanalysis were derived from cohort studies using survival analyses (n = 6 HR studies). When pooled, these cohort-based HRs suggested a reduced risk of incident dementia associated with allopurinol use; however, heterogeneity was substantial.
This heterogeneity was primarily attributable to a single highweight cohort reporting an effect estimate in the opposite direction. Studies employing case-control or nested casecontrol designs, which reported ORs rather than HRs, were excluded from HR pooling and addressed separately in the secondary analysis.
Allopurinol Dose: Dose-stratified analyses of HR-based studies indicated that higher average daily doses of allopurinol (approximately 300 mg/day; n = 2 HR studies: Singh 201830; Hong 2015) were associated with more consistent protective associations against dementia, with low heterogeneity.
In contrast, studies reporting lower average doses (approximately 200 mg/day; n = 4 HR studies: Min 2021; Singh 201831; Scheepers 2019; Kim 2023) exhibited greater variability, with pooled estimates approaching but not consistently reaching statistical significance. Differences in exposure duration and patient characteristics may partly explain this variability.
Geographic Region: When stratified by geographic region, Asian cohorts (Taiwan and Korea; n = 3 HR studies: Min 2021; Hong 2015; Kim 2023) demonstrated a more consistent protective association between allopurinol use and dementia risk, with moderate heterogeneity.
In contrast, Western cohorts (United States and Europe; n = 3 HR studies: Singh 201830; Singh 201831; Scheepers 2019)
showed greater heterogeneity and pooled estimates closer to the null. This pattern was largely influenced by one large U.S. cohort reporting an increased risk estimate.
Age Group: Age-stratified HR analyses indicated that patients younger than 65 years (n = 3 HR studies: Min 2021; Hong 2015; Scheepers 2019) experienced a clearer protective association between allopurinol use and incident dementia.
Among older adults (≥65 years) (n = 2 HR studies: Singh 201831; Kim 2023), pooled estimates were generally neutral and characterized by substantial heterogeneity, likely reflecting higher comorbidity burden, polypharmacy, and treatment channeling in older populations.
Follow-Up Duration: Studies with shorter to intermediate follow-up durations (<10 years; n = 3 HR studies: Singh 201830; Hong 2015; Kim 2023) consistently demonstrated a protective association between allopurinol use and dementia risk.
By contrast, studies with longer follow-up durations (≥10 years; n = 3 HR studies: Min 2021; Scheepers 2019; Singh 201831) showed attenuation of the protective effect toward neutrality. This may reflect time-varying exposure, declining adherence, or the accumulation of competing risks over prolonged follow-up.
Primary meta-analysis (hazard ratios)
HR-based results represent the primary analysis and form the basis of the main conclusion. Hazard ratios (HRs) were used as the primary effect measure because incident dementia is a time-to-event outcome. HRs account for differences in followup duration and censoring across cohorts, providing clinically interpretable estimates of dementia risk over time. Accordingly, we pooled adjusted HRs (95% CIs) reported from longitudinal analyses (e.g., Cox regression). Three studies were excluded from the HR-based synthesis because they reported odds ratios rather than hazard ratios or did not provide extractable survival estimates, as illustrated in Figure 2.
Only studies reporting time-to-event estimates from survival models were included. Studies reporting odds ratios (e.g., Engel et al., 201833; Chuang et al., 202134) and letter-based reports Lai et al., 202129 were excluded from this analysis and addressed separately
Visual inspection of the funnel plot based on adjusted hazard ratios (Figure 3) did not demonstrate clear asymmetry suggestive of small-study effects. However, interpretation is limited by the small number of HR-based studies and by between-study heterogeneity; therefore, publication bias cannot be reliably excluded. Formal statistical tests for funnel plot asymmetry were not performed because of limited power.
In the primary pooled HR analysis including all eligible HRbased studies, allopurinol exposure was associated with a non-significant reduction in incident dementia risk (pooled HR = 0.85, 95% CI 0.72–1.01), accompanied by substantial heterogeneity (I² = 89%). Because one large cohort study (Singh et al., 2018) reported an effect estimate in the opposite direction and contributed disproportionate statistical weight, a prespecified influence (sensitivity) analysis was conducted Figure 4.
After exclusion of this influential study, allopurinol exposure was associated with a statistically significant reduction in dementia incidence (pooled HR = 0.78, 95% CI 0.71–0.86), and heterogeneity decreased to a moderate level (I² = 43%). These findings indicate that the overall estimate is sensitive to the inclusion of a single high-weight study and suggest that residual confounding, treatment channeling, or differences in exposure definition may partly explain the observed heterogeneity. Accordingly, results from both the primary and sensitivity analyses are considered in interpretation.
Across individual studies, the majority reported hazard ratios below unity, indicating a neutral-to-protective association between allopurinol use and dementia risk, with Min et al. (2021) demonstrating the strongest protective estimate. Nevertheless, given the observational design of the included studies and the influence of individual large cohorts, causal inference remains limited. The funnel plot displays the log (HRs) plotted against their standard errors. Visual inspection suggests approximate symmetry; however, interpretation is limited by the small number of included studies and the presence of one large cohort with substantial statistical weight. Formal tests for funnel plot asymmetry were not performed due to insufficient power (n < 10). Accordingly, this figure should be interpreted as a qualitative assessment only Figure 5.
Secondary meta-analysis (odds ratios)
This secondary analysis included studies reporting adjusted odds ratios (ORs). ORs were pooled separately and were not combined with hazard ratios (HRs). Lai et al. (2021) is displayed descriptively but was not included in the pooled OR estimate.
One brief letter (Lai et al., 2021) reported an association between allopurinol use and dementia risk but was excluded from quantitative pooling because of limited methodological detail and publication format; the estimate is presented descriptively.
In Chuang et al. (2021), the allopurinol-specific estimate
derived from conditional logistic regression showed no significant association with dementia risk (adjusted OR 1.02, 95% CI 0.86–1.22).
Only studies reporting adjusted odds ratios (ORs) for allopurinol were eligible for pooling. Chuang et al. (2021) was a nested case–control study using conditional logistic regression; the OR shown corresponds specifically to allopurinol exposure, not other urate-lowering therapies. Lai et al. (2021) is presented descriptively and was not included in the pooled estimate. Results should be interpreted cautiously due to the limited number of OR-based studies44-48.
DISCUSSION
This updated meta-analysis synthesized observational evidence examining whether long-term allopurinol exposure is associated with incident dementia in patients with gout or hyperuricemia. In the primary HR-based analysis, allopurinol was not associated with an increased risk of dementia, indicating a neutral association overall. However, because one very large cohort study reported a directionally opposite estimate and contributed disproportionate statistical weight, a prespecified sensitivity analysis excluding this study was performed. In this sensitivity analysis, allopurinol exposure was associated with a statistically significant reduction in incident
dementia, and heterogeneity decreased to a moderate level. Taken together, these findings support a neutral association in the primary analysis, with evidence of a potentially protective effect in sensitivity analyses, while underscoring the inherent limitations of causal inference in observational research.
From a mechanistic perspective, the relationship between urate-lowering therapy and cognition is biologically plausible but complex. Uric acid may exert antioxidant effects, whereas xanthine oxidase activity contributes to oxidative stress and inflammatory signaling processes implicated in neurodegeneration. Allopurinol may therefore influence dementia pathways through competing mechanisms, namely reduction of serum urate versus inhibition of xanthine oxidase– derived oxidative stress. This duality may partly explain the variability observed across populations, age strata, and study designs.
Lai et al. reported a neutral association between allopurinol use and dementia risk. Because this study was published as a brief letter with limited methodological detail and did not report time-to-event estimates, it was presented descriptively and excluded from the pooled HR-based meta-analysis. Nevertheless, its findings were directionally consistent with the overall neutral-to-protective pattern observed across the included studies.
Implications for pharmacy practice: MTM and comprehensive medication reviews (CMRs)
These findings are clinically relevant because allopurinol is commonly initiated and then continued for many years, particularly in older adults with multimorbidity and polypharmacy. Pharmacists play a central role in medication therapy management and comprehensive medication reviews, including confirmation of indication and treatment goals, support for adherence, monitoring for adverse effects and drug–drug interactions, and coordination of dose titration with prescribers. The present results provide reassurance that longterm allopurinol therapy does not appear to increase dementia risk overall, supporting continued guideline-based use when clinically indicated and appropriately monitored.
Identification and monitoring of potential cognitive adverse effects
Although dementia outcomes in administrative datasets rely on diagnostic coding, routine pharmacy follow-up provides opportunities to detect early cognitive changes that are clinically meaningful regardless of causality. During refill encounters or medication reviews, pharmacists can screen for new memory complaints, confusion, reduced ability to manage medications, functional decline, or increased falls, and communicate concerns promptly to prescribers for formal evaluation. Integrating this vigilance into routine follow-up represents a pragmatic safety strategy in high-risk older adults.
Confounding by indication and comorbidity burden
The heterogeneity observed across studies likely reflects differences in baseline risk and treatment channeling. Patients receiving allopurinol may differ systematically from non-users in cardiovascular disease burden, chronic kidney disease, inflammatory status, frailty, healthcare utilization, and overall medication complexity—factors independently associated with dementia risk. Although most included studies adjusted for major confounders, residual confounding remains plausible, particularly for gout severity, duration of hyperuricemia, lifestyle factors, and adherence. These issues may be amplified in very large administrative cohorts, where modest residual bias can exert substantial influence on pooled estimates.
Dose optimization, deprescribing, and pharmacist–prescriber coordination (CKD focus)
Dose selection and titration are key pharmacy contributions, particularly in older adults and patients with CKD, where dose adjustments are frequently required. Pharmacists can support safe initiation using a start-low and titrate-slow approach, reinforce adherence, monitor renal function and tolerability, and coordinate dose adjustments with prescribers to achieve urate targets while minimizing toxicity. Deprescribing should be individualized and considered when the original indication is no longer present or when adverse effects outweigh benefits. Importantly, the current evidence does not support routine discontinuation of allopurinol based on cognitive concerns alone, instead favoring structured follow-up and shared decision-making.
Strengths, limitations, and future directions
Strengths of this study include synthesis of populationbased cohorts with extended follow-up and prioritization of adjusted hazard ratios to capture time-to-event dementia outcomes. Limitations include the observational nature of the evidence, heterogeneity in exposure definitions and outcome ascertainment, limited detail on dose and adherence, and the potential for residual confounding. Future studies incorporating richer clinical data should evaluate effect modification by age, CKD status, comorbidity burden, and treatment intensity to improve applicability to medication decision-making.
CONCLUSION
In conclusion, this meta-analysis suggests that long-term allopurinol use is not associated with an increased risk of dementia and may be associated with a reduced incidence of dementia in patients with gout or hyperuricemia. While the primary hazard ratio–based analysis indicated a neutral association overall, sensitivity analyses excluding a single influential cohort consistently demonstrated a statistically significant protective association, with most included studies reporting effect estimates below unity.
These findings support the interpretation that allopurinol therapy is cognitively safe and potentially protective, particularly in certain subgroups, such as younger patients, those receiving higher daily doses, and individuals with lower comorbidity burden. From a clinical perspective, the results provide reassurance for pharmacists and prescribers involved in long-term urate-lowering therapy that continuation of allopurinol is unlikely to confer cognitive harm and may confer benefit in selected populations.
Nevertheless, given the observational design of the included studies and the presence of heterogeneity and potential residual confounding, causal conclusions cannot be definitively established. Future prospective studies with standardized exposure definitions, detailed dose and duration assessment, and robust adjustment for comorbidity burden are warranted to confirm whether the observed protective association reflects a true neuroprotective effect of allopurinol.
Acknowledgments
The authors would like to acknowledge the financial support provided by the KAU Endowment (WAQF) at King Abdulaziz University, Jeddah, Saudi Arabia. The authors, therefore, acknowledge with thanks WAQF and the Deanship of Scientific Research (DSR) for technical and financial support.
Funding
The project was funded by KAU Endowment (WAQF) at King Abdulaziz University, Jeddah, Saudi Arabia. The authors, therefore, acknowledge with thanks WAQF and the Deanship of Scientific Research (DSR) for technical and financial support.
References
- Wu, A.H., et al., Relation of serum uric acid to cardiovascular disease. International Journal of Cardiology, 2016. 213: p. 4-7.
- Ndrepepa, G., Uric acid and cardiovascular disease. Clinica chimica acta, 2018. 484: p. 150-163.
- Euser, S.M., et al., Serum uric acid and cognitive function and dementia. Brain, 2009. 132(2): p. 377-382.
- Ruggiero, C., et al., Uric acid and dementia in community-dwelling older persons. Dementia and geriatric cognitive disorders, 2009. 27(4): p. 382-389.
- Latourte, A., et al., Uric acid and incident dementia over 12 years of follow-up: a population-based cohort study. Annals of the rheumatic diseases, 2018. 77(3): p. 328-335.
- Mazzali, M. et al., Elevated uric acid increases blood pressure in the rat by a novel crystal-independent mechanism. hypertension, 2001. 38(5): p. 1101-1106.
- Facts, A.S.D., and Figures, 2022 Alzheimer’s disease facts and figures. Alzheimers Dement, 2022. 18(4): pp. 700-789.
- Feig, D.I., D.-H. Kang, and R.J. Johnson, Uric acid and cardiovascular risk. New England Journal of Medicine, 2008. 359(17): p. 1811-1821.
- Zhang, W.-z., Uric acid en route to gout. Advances in Clinical Chemistry, 2023. 116: p. 209-275.
- Drivelegka, P., The population burden of gout and urate in Western Sweden. 2021.
- Cummings, J., et al., Alzheimer’s disease drug development pipeline: 2019. Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 2019. 5: p. 272-293.
- Choi, H.K., et al., Purine-rich foods, dairy and protein intake, and the risk of gout in men. New England Journal of Medicine, 2004. 350(11): p. 1093-1103.
- Nichols, E., et al., Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. The Lancet Public Health, 2022. 7(2): p. e105-e125.
- Van der Flier, W.M. and P. Scheltens, Epidemiology and risk factors of dementia. Journal of Neurology, Neurosurgery & Psychiatry, 2005. 76(suppl 5): p. v2-v7.
- Wang, S., The efficacy of febuxostat and allopurinol in the treatment of gout with hyperuricemia. Pakistan Journal of Pharmaceutical Sciences, 2018. 31.
- Vargas-Santos, A.B., et al., Association of chronic kidney disease with allopurinol use in gout treatment. JAMA internal medicine, 2018. 178(11): p. 1526-1533.
- Smalley, R.V., et al., Allopurinol: intravenous use for prevention and treatment of hyperuricemia. Journal of Clinical Oncology, 2000. 18(8): p. 1758-1763.
- Castrejon, I., et al., Safety of allopurinol compared with other urate-lowering drugs in patients with gout: a systematic review and meta-analysis. Rheumatology International, 2015. 35: p. 1127-1137.
- Hay, C.A., et al., Mortality in patients with gout treated with allopurinol: a systematic review and meta‐analysis. Arthritis Care & Research, 2021. 73(7): p. 1049-1054.
- Wang, M., et al., The major cardiovascular events of febuxostat versus allopurinol in treating gout or asymptomatic hyperuricemia: a systematic review and meta-analysis. Annals of Palliative Medicine, 2021. 10(10): p. 103270337-103210337.
- Fan, M., et al., Comparison of efficacy and safety of urate-lowering therapies for hyperuricemic patients with gout: a metaanalysis of randomized, controlled trials. Clinical Rheumatology, 2021. 40: pp. 683-692.
- Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta- Analyses: The PRISMA Statement. PLoS Med. 2009;6(7):e1000097.
- Methley, A.M., et al., PICO, PICOS, and SPIDER: a comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Services Research, 2014. 14(1): p. 1-10.
- Lo, C.K.-L., D. Mertz, and M. Loeb, Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Medical Research Methodology, 2014. 14: p. 1-5.
- Takkouche, B. and G. Norman, PRISMA statement. Epidemiology, 2011. 22(1): p. 128.
- Scheepers, L.E., et al., Urate and risk of Alzheimer’s disease and vascular dementia: a population-based study. Alzheimer’s & Dementia, 2019. 15(6): p. 754-763.
- Kim, J.H., et al., Impact of clinical association between gout and dementia: A nationwide population‐based cohort study in Korea. Arthritis Care & Research, 2023. 75(5): p. 1088-1094.
- Hong, J.-Y., et al., Gout and the risk of dementia: a nationwide population-based cohort study. Arthritis research & therapy, 2015. 17: p. 1-7.
- Lai, S.-W., C.-L. Lin, and K.-F. Liao, Association between allopurinol use and dementia in the elderly. The American Journal of Geriatric Psychiatry, 2021. 29(11): p. 1174-1175.
- Singh, J.A. and J.D. Cleveland, Comparative effectiveness of allopurinol versus febuxostat for preventing incident dementia in older adults: a propensity-matched analysis. Arthritis Research & Therapy, 2018. 20: p. 1-9.
- Singh, J.A. and J.D. Cleveland, Gout and dementia in the elderly: a cohort study of Medicare claims. BMC geriatrics, 2018. 18: p. 1-8.
- Min, K.H., et al., Association between gout and dementia in the elderly: a nationwide population-based cohort study. The American Journal of Geriatric Psychiatry, 2021. 29(12): p. 1177-1185.
- Engel, B., et al., Hyperuricemia and dementia—a case-control study. BMC Neurology, 2018. 18: p. 1-7.
- Chuang, T.-J., et al., Association Between Use of Anti-gout Preparations and Dementia: Nested Case-Control Nationwide Population-Based Cohort Study. Frontiers in Medicine, 2021. 7: p. 607808.
- MacIsaac, R.L., et al., Allopurinol and cardiovascular outcomes in adults with hypertension. Hypertension, 2016. 67(3): p. 535- 540.
- Chen, P.Y., et al., Association between colchicine use and the risk of dementia among patients with gout: A nationwide retrospective cohort study. International Journal of Rheumatic Diseases, 2024. 27(5): p. e15162.
- Ahn, J.K., Epidemiology and treatment-related concerns of gout and hyperuricemia in Koreans. Journal of Rheumatic Diseases, 2023. 30(2): p. 88-98.
- Molet-Benhamou, L., et al., Association between urate-lowering therapies and cognitive decline in community-dwelling older adults. Scientific Reports, 2022. 12(1): p. 15299.
- Lara, D.R., et al., Allopurinol for the treatment of aggressive behavior in patients with dementia. International clinical psychopharmacology, 2003. 18(1): p. 53-55.
- Lai, S.-W., et al., Allopurinol use and the risk of dementia: A meta-analysis of case-control studies. Medicine, 2022. 101(26): p. e29827.
- Morsy, A., et al., Functionalized allopurinols targeting amyloid-binding alcohol dehydrogenase rescue Aβ-induced mitochondrial dysfunction. ACS Chemical Neuroscience, 2022. 13(14): p. 2176-2190.
- Wei, S., Common Gout Medication May Help Reduce Risk for Neurodegenerative Diseases. Psychiatry Advisor, 2023: p. NA-NA.
- Cochrane Handbook for Systematic Reviews of Interventions (Version 6.3, 2022).
- Egger, M., Davey Smith, G., Altman, D. G. (2008). Systematic Reviews in Health Care: Meta-Analysis in Context. BMJ Books.
- Zhang, J., & Yu, K. F. (1998). What’s the Relative Risk? A Method of Correcting Odds Ratios in Cohort Studies of Common Outcomes. JAMA, 280(19): 1690–1691.
- Blettner, M., Sauerbrei, W., Schlehofer, B., Scheuchenpflug, T., & Friedenreich, C. (1999). Traditional reviews, meta-analyses, and pooled analyses in epidemiology. International Journal of Epidemiology, 28(1): 1–9.
- Bellou, V., Belbasis, L., Tzoulaki, I., Evangelou, E., & Ioannidis, J. P. A. (2016). Environmental risk factors and Alzheimer’s disease: An umbrella review of meta-analyses. PLoS Medicine, 13(6): e1002224.
- Goh, C., Banerjee, G., & Dutta, A. (2014). Uric acid and risk of dementia in the elderly: A meta-analysis. Journal of Alzheimer’s Disease, 42(4): 1235–1240.
