Morbidity and mortality rates in premature infants treated with aminoglycosides in neonatal intensive care units: A comparative study
Lujain Khalid Khan. PharmD, MSci, PhD Student at Department of Pharmacology and Toxicology-King Saud University, Senior Pharmacist at maternity and Children’s hospital, Makkah, Saudi Arabia 444204453@student.ksu.edu.sa
Walid A. Al-Qerem*. PhD, Associate Professor , Faculty of Pharmacy, Al-Zaytoonah University of Jordan, Amman, Jordan, waleed.qirim@zuj.edu.jo
Shakir D. AlSharari. PhD, Associate Professor, Department of Pharmacology and Toxicology, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh 11451, Kingdom of Saudi Arabia, sdalsharari@ksu.edu.sa
Fawaz Alasmari*. PhD, Associate Professor and Consultant, Department of Pharmacology and Toxicology, College of Pharmacy, King Saud University, P.O. Box 2457, Riyadh 11451, Kingdom of Saudi Arabia; King Salman Center for Disability Research, Riyadh 11614, Saudi Arabia. ffalasmari@ksu.edu.sa
DOI: https://doi.org/10.18549/PharmPract.2025.4.3342
Abstract
Objective: Aminoglycosides are used as first-line defense antibiotics in the Neonatal Intensive Care Unit (NICU) for the treatment of sepsis, meningitis, neonatal pneumonia, and endocarditis. However, they pose negative side effects such as ototoxicity, and nephrotoxicity, and the neuromuscular blockade effect is resulting from acetylcholine inhibition. In contrast to other antibiotics (such as vancomycin or meropenem), delayed weaning and weaning failure from artificial ventilation may be linked to neuromuscular blocking in neonates treated with aminoglycosides. However, the used dosing regimen and therapeutic guideline of aminoglycosides may provide optimal clinical outcomes. Methods: Data were obtained for 582 NICU patients with sepsis, birth asphyxia, respiratory distress syndrome, birth defects, infections or others, and who needed artificial ventilation. Included patients were divided into four groups: A, treated with amikacin; B, treated with gentamicin; C, treated with meropenem; and D, treated with vancomycin. The weaning duration, weaning failure rate, mortality rate, and length of hospital stay were compared. Results: Gentamicin showed the most positive effects in reducing the length of hospital stay and ventilation period of neonates with improved health conditions. In addition, the mortality rate was lowest in neonates treated with gentamicin as compared to other treatments. Conclusion: Our data elucidated that aminoglycosides, particularly gentamicin, with guidance was effective in improving the neonatal mortality and morbidity through reducing length of hospital stay and ventilation period without producing neuromuscular blocking action.
Keywords: Aminoglycosides; Meropenem; Vancomycin; NICU; Mortality; Morbidity; Ventilation; Length of stay
INTRODUCTION
closely related class of bactericidal antibiotics known as aminoglycosides is made up of an aminocyclitol ring that is joined to amino sugars by glycosidic linkage. They are derived from gram-positive bacteria belonging to the general Micromonospora (gentamicin) and Streptomyces (tobramycin)1. As broad-spectrum antimicrobial agents, aminoglycosides are effective against gram-negative aerobic bacteria and Staphylococcus aureus2. Despite the development of third- and fourth generation cephalosporins and other such new antibiotics, older aminoglycosides such as gentamicin, tobramycin, and amikacin are still important for treating serious neonatal infections3-5. Indeed, gentamicin is both the most common aminoglycoside in the UK and the most commonly used antibiotic in neonatal units6. As a first-line treatment, gentamicin is often used when treating serious bacterial infections in neonates, and as an experimental treatment, it is usually employed in combination with another antibiotic, such as penicillin7. In pediatric medicine, gentamicin is used for a variety of indicators, including septicemia, meningitis, urinary tract infections, gastrointestinal tract infections, respiratory tract infections, bone infections, infections, eye infections, subcutaneous tissue infections or peritonitis of peritoneal dialysis8.
Despite their widespread use, aminoglycosides are not highly safe; in particular, studies have demonstrated these antibiotics to have high rates of renal toxicity9-12, and the potential to induce irreversible hearing loss13. At the molecular level, both the antibacterial and toxic effects of aminoglycosides are attributable to hyperactivation of cellular respiration, which results in oxidative stress, and perturbation of the translation machinery, which produces mistranslated and misfolded proteins14. There is evidence that structural modification of aminoglycoside antibiotics can reduce their detrimental effects15; however, the pharmaceutical industry has not made significant advancements in this direction. Pediatric studies have indicated that lowering dose frequency and increasing dose amount may reduce toxicity16,17. In the absence of safer drug development, it is necessary to comprehensively characterize the adverse effects of aminoglycosides and determine approaches for reducing the risk of those effects. However, adhering to the therapeutic guideline and appropriate dosing regimens can play major role in reducing the occurrence of toxicity and effects significantly in neonates. Here we report clinical evidence of aminoglycoside efficacy in neonates, including mortality and morbidity rates, along with differences between amikacin and gentamicin.
Acute kidney injury (AKI) can also be linked to antibiotic therapy, particularly higher vancomycin trough concentrations; although this risk is generally low, the incidence of AKI in neonates treated with vancomycin is 2.7%18. Amikacin does not significantly impact kidney function as measured by serum creatinine level, but it does cause a temporary rise in urinary N-acetyl-β-D-glucosaminidase, indicative of minor and reversible tubular damage19,20. Meanwhile, gentamicin treatment leads to higher serum creatinine, which is in turn associated with increased urine biomarkers KIM-1, GSTA1- 1, and GSTP1-1; hence, these markers could help in early detection of AKI in neonates in the NICU21. No link between the nephrotoxicity or chronic kidney disease and meropenem use has been documented.
Although meropenem has no reported association with hearing and kidney injury, Cohen-Wolkowiez et al. (2012) found that among 200 infants who received intravenous meropenem, 10 (5%) experienced seizures22. However, electroencephalography conducted on 6 of these subjects confirmed only one seizure22. Furthermore, one subject who had a seizure was not in fact on the study drug that day22. Interestingly, the average predicted Cmax of meropenem was similar in infants both with and without seizures: 57.18 mg/L (±13.50) vs. 53.12 mg/L (±5.08), with a P-value of 0.2422. Gentamicin may have a neuromuscular blocking effect, as a case report by Mitali Sahni et al. (2015) found that using gentamicin alongside vecuronium enhances neuromuscular blockade (NMB) 23. This study reported that the toxic concentrations of the serum gentamicin was reached (peak: 9.8 μg/mL, trough: 2.2 μg/mL). They also suggested that in patients with renal failure, prolonged use of vecuronium in combination with gentamicin and steroids could lead to extended and persistent NMB23.
In addition, with amikacin, neuromuscular blockade can be occurred. However, amikacin is usually used in infants when no alternative is available because they have a larger volume of distribution, leading to prolonged systemic circulation of the drug24. However, amikacin was used for the treatment of early and late onset neonatal sepsis25,26. A case series by Varia et al. (2017) documented a night dose of amikacin sulfate to be followed by severe adverse reactions in five preterm low-birthweight newborns, including lethargy and bluish discoloration of the body, which might be attributed to neuromuscular blockade effects of amikacin27. However, this study highlighted that it is impossible to determine the precise cause of these adverse events because it only involved a single episode of incidents and there was no clarification for drug preparation or administration. Amikacin was found to induce less potential of neuromuscular blockade compared to other aminoglycosides28. Gentamicin can cause neuromuscular blockade at high and accumulative doses29. Like gentamicin, vancomycin is also reported to enhance the effects of neuromuscular-blocking agents, while meropenem has not been associated with neuromuscular blockade.
Recent years have seen significant improvement in the survival of newborns, especially premature babies, on account of advances in the prevention of preterm poisoning. However, while fewer children now die due to respiratory failure, infection has become an increasing cause of mortality and morbidity. In particular, infection due to gram-negative bacteria accounts for up to 25% of all sepsis episodes in neonatal units and often affects the smallest and most premature babies6. Our clinical study was conducted to explore the effects of aminoglycosides on improving the morbidity and mortality through decreasing the weaning duration as compared to meropenem or vancomycin in neonates. Our hypothesis is to evaluate the risk against benefit of using aminoglycoside in neonatalassociated infection in the Neonatal Intensive Care Unit (NICU) with artificial ventilation when compared with other antibiotic groups commonly used in neonates according to type and severity of infection and hospital antibiogram, through evolution of mortality, morbidity rate, weaning duration, and weaning failure rate to evaluate the neuromuscular blocking exert clinical toxicology and manifestation on the respiratory system of neonates treated with the aminoglycoside.
MATERIALS AND METHODS
Setting and study population
A retrospective cohort study was conducted at the NICU of the Maternity and Children Hospital in Mecca, Kingdom of Saudi Arabia, covering the period of 2018-2024. In total, the study enrolled 582 patients at gestational age of 6-8 months diagnosed with sepsis, meningitis, and respiratory distress syndrome who were put on artificial ventilation and treated with intravenous (IV) aminoglycoside (gentamicin or amikacin) or non-aminoglycoside (meropenem) antibiotics. Compliance with the treatment regimen was ensured via medication administration sheet, nurse endorsement, physician order, and the HIS system.
Ethical consideration
Institutional Review Board (IRB) of the Local Committee for Research Ethics at the Health Sector of Makkah Al-Mukarramah Region in Saudi Arabia issued its approval to the current study (H-02-K-076-0724-1145). The research study complies with the IRB Committee’s criteria as well as the Declaration of Helsinki of 1975 and later amendments. IRB committee waived the requirement for obtaining informed consents. The committee approved that this study is qualified for exemption meaning that it has no more than minimal risk.
Inclusion criteria
Neonate with artificial ventilation, treated with aminoglycoside (amikacin or gentamicin) or non -aminoglycoside antibiotics (vancomycin or meropenem) following the therapeutic guideline, no disorder affecting the neuromuscular system (such as Duchenne muscular dystrophy), no metabolic syndrome, and not treated with a neuromuscular blocking agent. Propofol was used in some cases for general anesthesia.
Exclusion criteria
All neonates without artificial ventilation, diagnosed with a genetic disorder affecting the neuromuscular or metabolic system, having a monitoring or administration medication error for the rational use of aminoglycoside, lacking trough aminoglycoside levels in their file, or who experienced renal or liver impairments. The kidney and liver functions tests, including creatinine, alanine aminotransferase and aspartate aminotransferase levels in infants were within normal range.
Groups classification
A retrospective study was performed, and parameters of interest were collected and analyzed. The clinical study was designed as follows: Group A: 151 patients treated with amikacin and ampicillin, with amikacin target trough level (less than 5 mg/L) checked every 3-4 days. Group B: 150 patients treated with gentamicin and ampicillin, with gentamicin target trough level (less than 1 mg/L) checked every 3-4 days. Group C: 130 patients treated with antibiotics other than aminoglycosides (meropenem and a cephalosporin agent except ceftriaxone) No trough level or peak are required. Group D: 151 patients treated with antibiotics other than aminoglycosides (vancomycin and other cephalosporin except ceftriaxone) No trough level obtained before the fourth dose by 30 minutes. All groups were treated with ampicillin and gentamicin for 24-48 hours till the outcome of the culture and antibiotic changed according to the recommended guideline by the MOH and culture sensitivity, where gentamicin is used as the first line therapy. Mortality rates, weaning failure time, length of stay, and the number of ventilation techniques applied were determined and compared between the studied four groups. A flowchart revealing the recruitment process of participants for analysis is illustrated in figure 1.
Statistical analysis
SPSS version 26 was used to perform statistical analyses for continuous and categorical variables. Continuous variables were presented as medians and 25-75 percentiles, while categorical variables were presented as frequencies and percentages. The normality of length of the stay and length of ventilation were assessed by examining Q-Q plots, and as the data was not normally distributed, nonparametric tests were conducted and two quantile regression analyses were carried out to assess the association between these two variables and various variables including gender, primary diagnosis and medications (Amikacin, Gentamicin, Meropenem and Vancomycin). A Binary logistic regression was performed to assess the association of the predictors (gender, primary diagnosis and medications) with improvement vs. mortality. Multicollinearity between the different predictors were evaluated by computing VIF values and all the values were less than 3. Kruskal-Wallis followed by Dunn’s multiple comparison test was used to further assess the significance of premature infants’ weight and hospitalization stay as well as ventilation lengths and failure rates between groups in combination or separation of improved and dead infants. The significance level was set at a threshold of p < 0.05. The appropriate target participants were calculated based on the population size with 95% confidence interval of each group in the hospital as follow; amikacin (n= 145), gentamicin (n = 149), meropenem (n= 113) and vancomycin (n= 116).
RESULTS
Socio-demographic characters
Table 1. depicts the Socio-demographic characters for the 582 premature infant patients (55.4% males). Amikacin, Gentamicin, Meropenem and Vancomycin were given to 151, 150, 130 and 151 neonates respectively. The median length of ventilation was 12 (3-33) days, while the median length of stay was 23.26 (10.8-41.76) days. Moreover, (65.6%) of the premature infants’ conditions improved. We also investigated the significant differences of the baseline of the body weight between the four groups. The statistical analysis did not reveal any significant changes in the body weight between the groups (Figure 2).
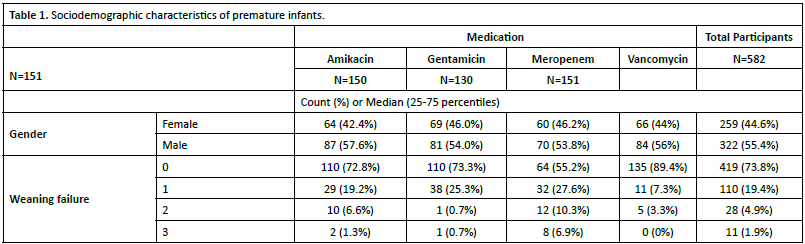
Identifying the variables significantly associated with premature infants’ hospitalization and ventilation lengths
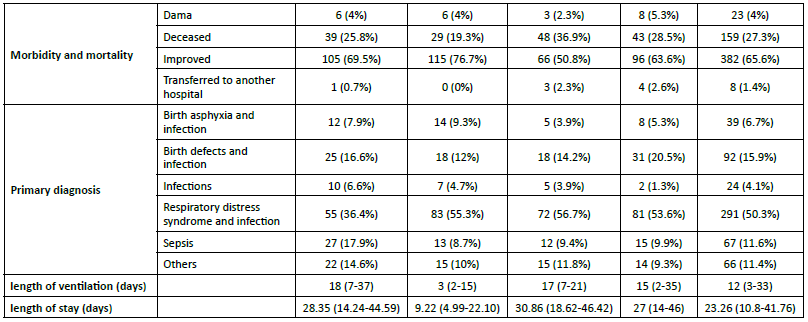
Two quantile regression models were applied to identify the variables significantly associated with premature infants’ hospitalization and ventilation lengths (Table 2). The results showed that premature infants who had been given gentamicin had a significantly shorter length of stay (Coefficient =-15.730, 95%CI (-21.132,-10.328), p<0.001), and shorter length of ventilation (Coefficient =-13.000, 95%CI (-18.007, -7.993), p<0.001) when compared with those who had been given vancomycin. Moreover, infants who had respiratory distress syndrome had significantly longer length of stay when compared to patients with sepsis (Coefficient =7.400, 95%CI
We further compared the premature infants’ hospitalization and ventilation lengths as well as number of ventilation failures between the four treatment groups using Kruskal-Wallis test. The statistical analysis showed significant differences in premature infants’ hospitalization lengths in total (p< 0.0001), improved (p< 0.0001), and dead infants (p< 0.001). Dunn’s multiple comparison revealed that gentamicin treated infants had significant reduction in hospitalization lengths as compared to other three treatment groups (Figure 3A). In addition, similar results were observed in improved infants, moreover, meropenem group had higher length of stay compared to amikacin group (Figure 3B). However, in dead
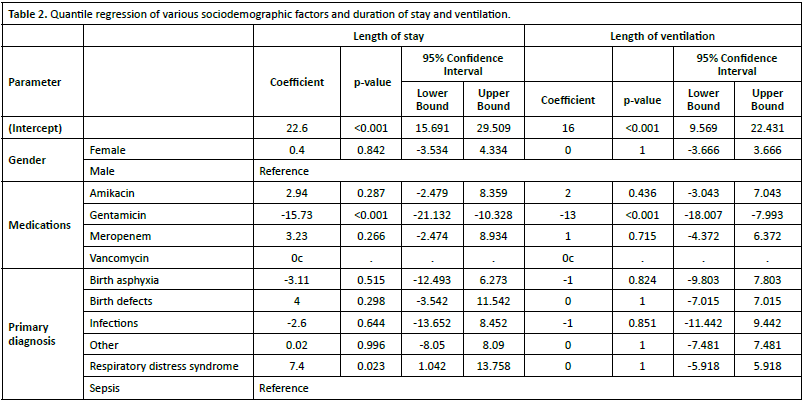

Figure 1. A flowchart revealing the recruitment process of participants for analysis. DAMA, discharge against medical advice
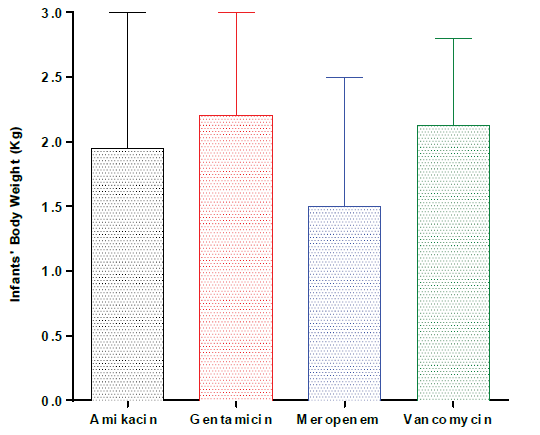
Figure 2. Infants’ body weight in kilogram (Kg). Amikacin, n= 94, Gentamicin, n=84, Merpenem, n= 92 and vancomycin, n= 105. Data is shown as median with interquartile range.
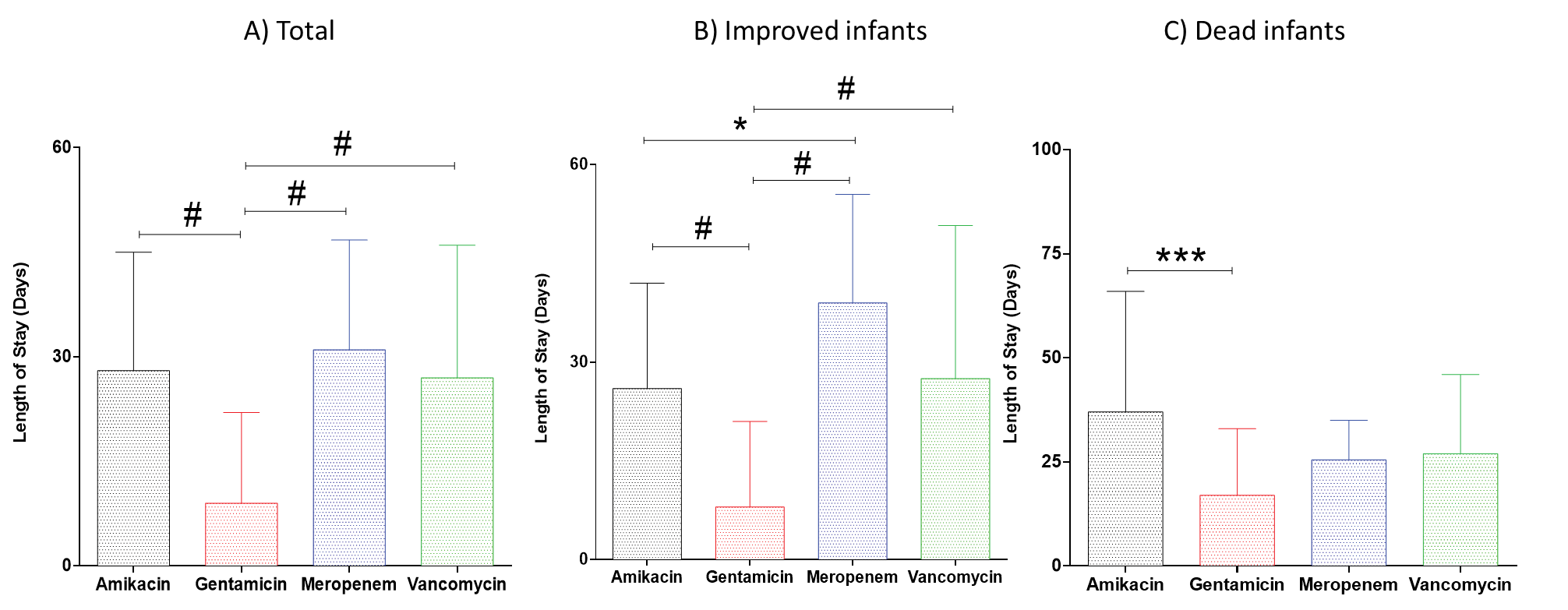
Figure 3. Effects of amikacin, gentamicin, meropenem and vancomycin on infants’ hospitalization lengths. *P< 0.05; ***P< 0.001; #P< 0.0001. Data is shown as median with interquartile range. infants, the length of hospitalization was only significantly higher in amikacin treated infants as compared to gentamicin treated infants (Figure 3C).
Kruskal-Wallis test showed significant differences in premature infants’ ventilation lengths in total (p< 0.0001), improved (p< 0.0001), and dead infants (p< 0.0001). Dunn’s multiple comparison revealed that gentamicin treated infants had significant reduction in ventilation lengths as compared to other three treatment groups (Figure 4A). In addition, similar results were observed in improved infants (Figure 4B). However, in dead infants, the length of ventilation was significantly higher in amikacin treated infants as compared to gentamicin or meropenem treated infants, and the length of ventilation was higher in vancomycin group compared to gentamicin group (Figure 4C).
Kruskal-Wallis test showed significant differences in premature infants’ ventilation failures in total (p< 0.0001), improved (p< 0.0001), and dead infants (p< 0.05). Dunn’s multiple comparison revealed that gentamicin, vancomycin and amikacin treated infants had significant lower in number of ventilation failures as compared to meropenem group, moreover, vancomycin group had also lower ventilation failures compared to amikacin and gentamicin groups (Figure 5A). In addition, in improved infants, meropenem had higher ventilation failures than the other three treatment groups (Figure 5B). However, in dead infants, the number of ventilation failures were significantly higher in meropenem treated infants as compared to infants treated with vancomycin (Figure 5C).
Identifying the variables significantly associated with patient improvement
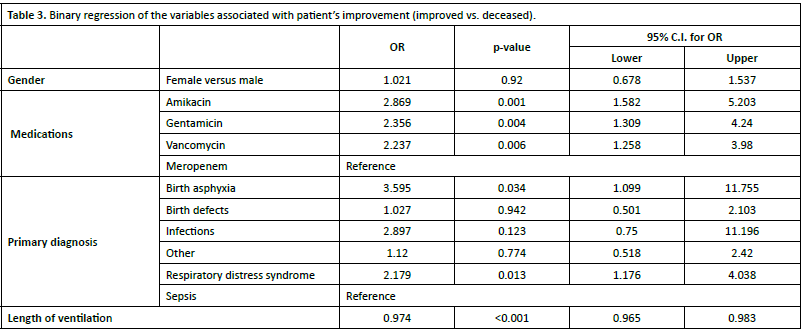
A binary regression model was applied to identify the variables significantly associated with patient improvement (displayed in Table 3). The results showed that the premature infants who received been Amikacin. Gentamicin or Vancomycin had significantly higher odds of being in the improvement group when compared with those who had been given Meropenem (OR =2.869, 95%CI (1.582-5.203), p=0.001; OR =2.356, 95% CI (1.309-4.240); p=0.004, and OR =2.237, 95% CI (1.258-3.980), p=0.006 respectively.). Moreover, patients with birth asphyxia had significantly higher odds of improvement than infants with sepsis (OR =3.595, 95%CI (1.099-11.755), p=0.034). Finaly, longer length of ventilation was associated with declined odds of improvement (OR =0.974, 95%CI (0.965-0.983), p<0.001).
DISCUSSION
In this study of 582 neonates who received antibiotics (amikacin, gentamicin, vancomycin or meropenem). These neonates were investigated for mortality and hospital stay as well as ventilation duration and failures. Concerning the mortality outcomes of participants, treatment with gentamicin demonstrated a good rate of improved health. Among patients receiving amikacin,
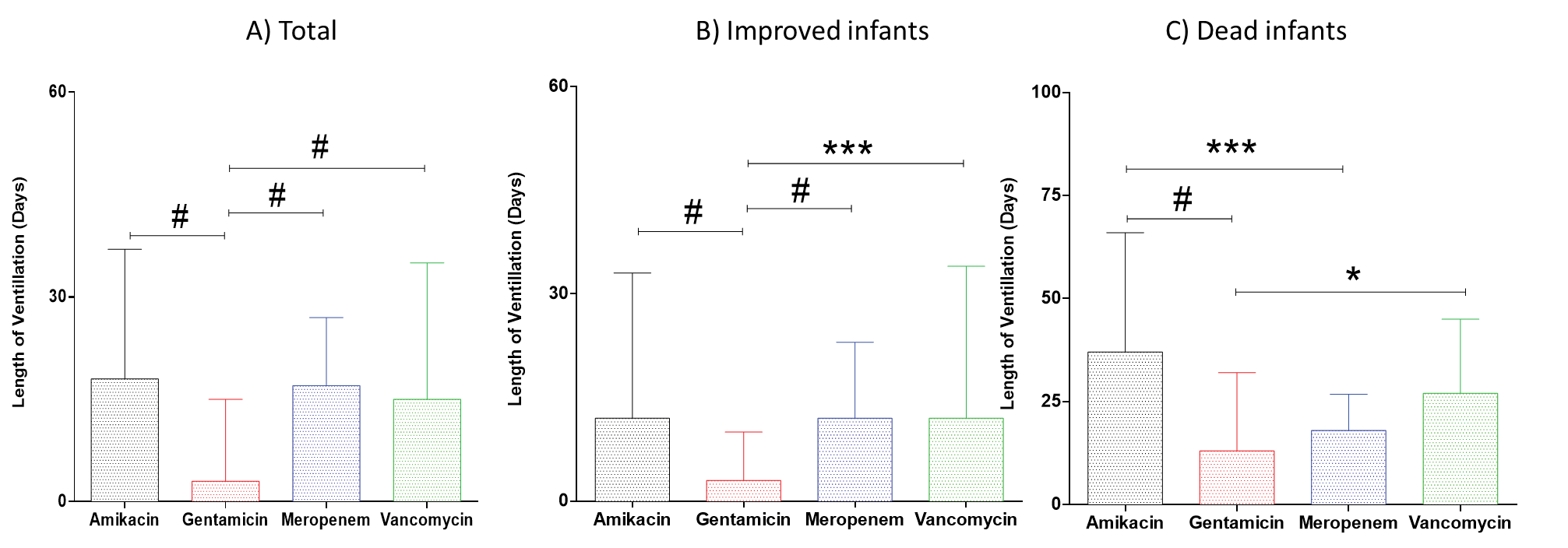
Figure 4. Effects of amikacin, gentamicin, meropenem and vancomycin on infants’ ventilation lengths. *P< 0.05; ***P< 0.001; #P< 0.0001. Data is shown as median with interquartile range
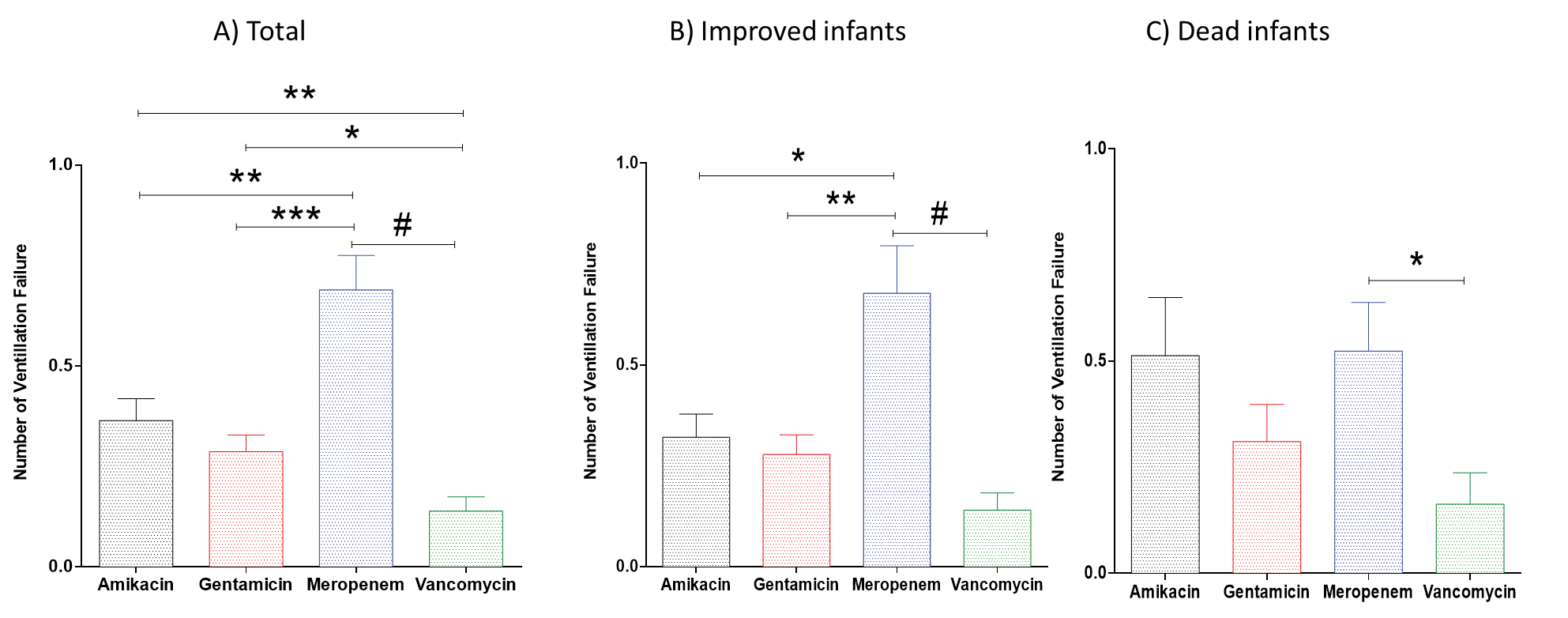
Figure 5. Effects of amikacin, gentamicin, meropenem and vancomycin on number of ventilation failures. *P< 0.05; **P< 0.01; ***P< 0.001; #P< 0.0001. Data is shown as median with interquartile range.
improvement was seen slightly less than with gentamicin. Of those treated with meropenem, improvement was the lowest rate among the tested antibiotics; at the same time, the number of deceased patients was highest. Vancomycin had the highest rate of discharge against medical advice (DAMA). Regarding weaning failure in relation to antibiotic treatment, the lowest rate was observed for vancomycin. The groups receiving gentamicin and amikacin exhibited moderate rates of failure, and did not differ significantly from one another. Those receiving meropenem had the highest failure rate. Regarding length of ventilation, patients who received gentamicin had the shortest median duration of ventilation, while those treated with amikacin had the longest. When considering hospital stay, the shortest median stay was observed for patients treated with gentamicin, while the highest was observed in meropenem group.
It is well-established in the literature that different antibiotics have distinct potential repercussions when applied to the treatment of infants and neonates30-32. A prior study investigating the morbidity associated with commonly-used antibiotics in neonates found that vancomycin use in neonates was associated with ototoxicity, as evidenced by a 22% otoacoustic emissions (OAE) failure rate in patients receiving vancomycin without gentamicin33. Conversely, gentamicin use did not correlate with increased risk of ototoxicity but was rather associated with a statistically significant decrease in OAE failure rate, with the lowest rate of 4% observed in those receiving gentamicin alone33, however gentamicin was found to cause ototoxicity in other cases. Meanwhile, amikacin trough concentrations of 10 μg/mL or higher in low-birthweight infants significantly elevate the risk of ototoxicity20, although in 35 neonates treated with amikacin, only one case of mild hearing loss was reported, which could not be definitively linked to the treatment19. More studies are needed to further confirm the link of these antibiotics exposure and the ototoxicity occurrence and severity.
Another perennial concern of antibiotic use is resistance. A study of 253 neonates found 101 (39.9%) to be colonized with vancomycin-resistant enterococci (VRE). Of these, 59 new cases were detected during the first nine weeks of the study period, among which molecular analysis revealed one predominant clone34. By weeks 10-12, no new cases of VRE colonization were found; however, a second wave erupted at week 13, resulting in 42 new cases and multiple clones34. In that study, 33 (40.2%) babies also became colonized with VRE, but the colonization rate decreased following implementation of infection control measures34. Similarly, a study involving 1,320 infants discharged from the NICU found 9% (119 infants) to be colonized with antimicrobial-resistant gram-negative bacteria (GNB)35. This included the 3.5% of cases with gentamicin resistance and the 1.7% with merpenem resistance35. Prolonged treatment with broad-spectrum antibiotics, specifically ≥10 days of treatment with third- or fourth-generation cephalosporins or meropenem, has been linked to colonization with gentamicin-resistant GNB35. Similarly, treatment with meropenem for ≥10 days is associated with colonization of carbapenem-resistant GNB35. That study also reported female sex and extended treatment with meropenem as related to presence of carbapenemresistant GNB in infants. However, it was found most infants who received prolonged (≥10 day) gentamicin or broadspectrum antibiotic treatment to not show positive culture results, with only 16% (14 out of 87) developing healthcareassociated infections resistant GNB35.
Another case reports study reviewed cultures positive for Escherichia coli isolated in neonatal sepsis and found a 12.9% incidence of gentamicin resistance36. Resistance rates vary by situation, but gentamicin-resistant E. coli is generally a rare cause of neonatal sepsis; nonetheless, it should be considered for patients whose condition worsens despite receiving empiric antibiotics36. The bacterial resistance plays a significant role in drugs choice for either treatment or prophylaxis of bacterial infections37. In the current study, the distributions of infections or sepsis treatment as a primary diagnosis indicate that amikacin is more widely applicable to a variety of infections. However, concerning resistance to amikacin, switching to amikacin in a neonatal unit was seen to trigger an outbreak of amikacin-resistant Serratia species, although these remained susceptible to gentamicin38. In the year following the switch, the frequency of amikacin resistance in nosocomial gramnegative infections rose significantly from 7.6% to 27.7% (P < 0.001), while the frequency of gentamicin resistance slightly decreased from 71.2% to 60.2% (P = 0.07) (38). Additionally, the rise in amikacin resistance among other gram-negative bacilli continued for over a year after amikacin became the only aminoglycoside used
Previous studies have shown amikacin to be effective against gram-negative bacteria, particularly resistant strains such as carbapenem-resistant Klebsiella pneumoniae (CRKP) 39,40. However, its use in CRKP infections has also been associated with higher mortality rates, especially when the minimum inhibitory concentration (MIC) is elevated (P = 0.019) 41. Studies further support that in patients with severe infections, usage of aminoglycosides was more prevalent in the mortality group (86.4%) compared to the survival group (61.1%), underscoring its potential risks41. Amikacin is also commonly used in combination therapies to treat meningitis caused by gram-negative bacteria like Klebsiella and E. coli, in which cases mortality rates range between 20 and 30%32. Therapeutic drug monitoring (TDM) has proven crucial in optimizing amikacin therapy, as improvement of TDM practices was found to lead to significant reduction in subtherapeutic peak levels and toxic trough levels42,43. This improvement was reflected in reduced neonatal sepsis mortality rates, which dropped from 45% in 1998 to 35% in 200042. Nonetheless, the association of amikacin with severe outcomes in certain infections highlights the importance of careful dosing and patient selection particularly in neonate populations44.
Compared to amikacin, gentamicin exhibits a more favorable safety profile. Gentamicin is frequently combined with ampicillin, particularly in treating neonatal infections, where it has been shown to reduce mortality compared to other regimens45. This study reported that elevated MIC values was found to reduce its effectiveness, but no significant association with mortality was reported (P = 0.168). Much as with amikacin, however, TDM has played a critical role in improving the safety and efficacy of gentamicin. Significant reductions in subtherapeutic and toxic levels were observed in 1999 and 2000 compared to 1998 (P < 0.05 for 1999, P < 0.001 for 2000), and this optimization likely contributed to decreased neonatal sepsis mortality rates42. Gentamicin is not without potential adverse effects, however, as it has been linked to increased incidence of neonatal necrotising enterocolitis, particularly when used in combination with vancomycin46. However, the combination of gentamicin with ampicillin demonstrated a lower mortality risk compared to ampicillin with cefotaxime in neonates45. Our data suggest that patients who received gentamicin had the shortest median duration of ventilation,
while those treated with amikacin had the longest, and the use of these two medications was associated with highest improved health conditions and lowest mortality rates in infants as compared to the other two antibiotics.
When treating gram-positive infections, vancomycin is a cornerstone antibiotic, including for resistant pathogens like methicillin-resistant Staphylococcus aureus, however, vancomycin resistant Enterococci was also well reported47,48. Even with this widespread use, especially in co-infections with CRKP, no statistically significant association of vancomycin with mortality has been observed (P = 0.178)41. However, toxic trough levels have become an increasing concern, rising from 25% in 1998 to 39% in 200042. These findings highlight the need for enhanced TDM practices to ensure optimal dose and minimize toxicity49. In pediatric with gram-positive bacteria, vancomycin at trough of 10-15 mg/L demonstrates good efficacy, with low mortality rates50; however, its utility against gram-negative pathogens remains limited. In addition, as mentioned above, its combination with gentamicin has been associated with increased risk of enterocolitis46. Meropenem stands out as the most effective option among the antibiotics discussed, particularly for gram-negative infections. Additionally, its effectiveness is well-documented in neonatal meningitis caused by resistant gram-negative pathogens such as extended-spectrum β-lactamase-producing Escherichia coli in infants51. While elevated MIC values are associated with increased mortality, appropriate use ensures superior outcomes compared to other regimens41. As best we are able to determine, specific TDM data is not currently available for meropenem in our study. The therapeutic management guidelines suggest the use of aminoglycosides for treatments and prevention of variety types of infections particularly in infants’ cases without complications such as resistance, which explain the effectiveness of gentamicin in improving the health status of infants via measuring the mortality and hospital length of stay and ventilation duration.
CONCLUSION
In cases without drug resistance, gentamicin was found to be effective and yields favorable outcomes as compared to other antibiotics. Most patients in thegentamicin group experienced weaning failure for only one day or none, whereas meropenem group, for example, struggled with weaning for multiple days. Additionally, patients who received gentamicin had both the shortest median ventilation period and the shortest median length of hospital stay. Regarding mortality, gentamicin was associated with the lowest death rate. Together, these observations suggest that gentamicin is the most effective option for enhancing patient outcomes, and hence a preferable treatment choice. For complicated cases, optimal treatment depends on the specific diagnosis as well as the presence of resistant strains of bacteria or any additional complications that may arise.
Amikacin was used for the treatment of sepsis, infections, and other conditions, and as prophylaxis for birth defects and asphyxia. Vancomycin was used mainly as prophylaxis in birth defects and respiratory distress syndrome. Meropenem should be reserved for cases involving gram-negative multi-resistant bacteria, as it exhibited the highest weaning failure rate as well as the longest length of hospital stay. We recommend that gentamicin be the first choice for treating mild to moderate cases of infection and for preventing the occurrence of infections in neonates. Our study indicated that for infections involving resistant bacteria, alternative therapies such as amikacin, vancomycin, or meropenem may be considered on a case-by-case basis. Future pharmaceutical industry research should focus on the physiochemical properties of gentamicin to innovate new compounds that offer better clinical outcomes alongside optimal safety and pharmacokinetic profiles.
This study was limited by the lack of certain data, such as gestational neonatal age, and assessment of infection severity, which is crucial for some illnesses such as sepsis. Additionally, the study did not report the type of organism, nor type of birth malformation such as patent ductus arteriosus. However, the culture sensitivity testing was the major factor classify the participants into four groups. Therefore, we believe that the data can be considered by health policy and therapeutic guidelines makers.
AUTHORS CONTRIBUTION
Conceptualization, W.A., F.A.; methodology, L.K.K., W.A., S.D.A., and F.A.; validation, L.K.K., W.A., and F.A.; formal analysis, L.K.K., W.A., and F.A.; investigation, W.A., F.A.; resources, W.A., F.A.; data curation, L.K.K., W.A., S.D.A., and F.A.; writing—original draft preparation, L.K.K., writing—review and editing, L.K.K., W.A., S.D.A., and F.A; supervision, F.A.; funding acquisition, F.A. All authors have read and agreed to the published version of the manuscript.
CONFLICTS OF INTEREST
Authors declare no conflict of interest.
FUNDING
The authors extend their appreciation to the King Salman center For Disability Research for funding this work through Research Group no KSRG-2024-156.
References
- Benveniste R, Davies J. Structure-activity relationships among the aminoglycoside antibiotics: role of hydroxyl and amino groups. Antimicrobial agents and chemotherapy. 1973;4(4):402-9.
- Young LS, Hewitt WL. Activity of five aminoglycoside antibiotics in vitro against gram-negative bacilli and Staphylococcus aureus. Antimicrobial Agents and Chemotherapy. 1973;4(6):617-25.
- Donowitz LG. Nesocomial infection in neonatal intensive care units. American journal of infection control. 1989;17(5):250-7.
- Hollander EM, van Tuinen EL, Schölvinck EH, Bergman KA, Bourgonje AR, Gracchi V, et al. Evaluation of Dosing Guidelines for Gentamicin in Neonates and Children. Antibiotics. 2023;12(5):810.
- Hartman SJ, Orriëns LB, Zwaag SM, Poel T, de Hoop M, de Wildt SN. External validation of model-based dosing guidelines for vancomycin, gentamicin, and tobramycin in critically ill neonates and children: a pragmatic two-center study. Pediatric Drugs. 2020;22:433-44.
- Muller-Pebody B, Johnson A, Heath P, Gilbert R, Henderson K, Sharland M, et al. Empirical treatment of neonatal sepsis: are the current guidelines adequate? Archives of Disease in Childhood-Fetal and Neonatal Edition. 2011;96(1):F4-F8.
- Camacho-Gonzalez A, Spearman PW, Stoll BJ. Neonatal infectious diseases: evaluation of neonatal sepsis. Pediatric Clinics. 2013;60(2):367-89.
- Chen C, Chen Y, Wu P, Chen B. Update on new medicinal applications of gentamicin: evidence-based review. Journal of the Formosan Medical Association. 2014;113(2):72-82.
- Mingeot-Leclercq M-P, Tulkens PM. Aminoglycosides: nephrotoxicity. Antimicrobial agents and chemotherapy. 1999;43(5):1003-1010
- Tulkens PM. Nephrotoxicity of aminoglycoside antibiotics. Toxicology letters. 1989;46(1-3):107-23.
- Le TA, Hiba T, Chaudhari D, Preston AN, Palowsky ZR, Ahmadzadeh S, et al. Aminoglycoside-related nephrotoxicity and ototoxicity in clinical practice: a review of pathophysiological mechanism and treatment options. Advances in therapy. 2023;40(4):1357-65.
- Rosenberg CR, Fang X, Allison KR. Potentiating aminoglycoside antibiotics to reduce their toxic side effects. PLoS One. 2020;15(9):e0237948.
- Selimoglu E. Aminoglycoside-induced ototoxicity. Current pharmaceutical design. 2007;13(1):119-26.
- Webster CM, Shepherd M. A mini-review: environmental and metabolic factors affecting aminoglycoside efficacy. World Journal of Microbiology and Biotechnology. 2023;39(1):7.
- Jospe-Kaufman M, Siomin L, Fridman M. The relationship between the structure and toxicity of aminoglycoside antibiotics. Bioorganic & medicinal chemistry letters. 2020;30(13):127218.
- Kotze A, Bartel P, Sommers DK. Once versus twice daily amikacin in neonates: prospective study on toxicity. Journal of paediatrics and child health. 1999;35(3):283-6.
- Lundergan FS, Glasscock B, F G, Kim EH, Cohen RS. Once-daily gentamicin dosing in newborn infants. Pediatrics. 1999;103(6):1228-
- Bhargava V, Malloy M, Fonseca R. The association between vancomycin trough concentrations and acute kidney injury in the neonatal intensive care unit. BMC pediatrics. 2017;17:1-6.
- Parini R, Rusconi F, Cavanna G, Vigliani E, Cornacchia L, Assael B. Evaluation of the renal and auditory function of neonates treated with amikacin. Developmental pharmacology and therapeutics. 1982;5(1-2):33-46.
- Endo A, Nemoto A, Hanawa K, Maebayashi Y, Hasebe Y, Kobayashi M, et al. Relationship between amikacin blood concentration and ototoxicity in low birth weight infants. Journal of Infection and Chemotherapy. 2019;25(1):17-21.
- Jansen D, Heemskerk S, Koster-Kamphuis L, Bouw T, Van Heijst A, Pickkers P. Urine biomarkers for gentamicin-induced acute kidney injury in the nicu. Pediatric Research. 2011;70(5):764-.
- Cohen-Wolkowiez M, Poindexter B, Bidegain M, Weitkamp J-H, Schelonka RL, Randolph DA, et al. Safety and effectiveness of meropenem in infants with suspected or complicated intra-abdominal infections. Clinical infectious diseases. 2012;55(11):1495-
- Sahni M, Richardson CJ, Jain SK. Sustained neuromuscular blockade after vecuronium use in a premature infant. American Journal of Perinatology Reports. 2015;5(02):e121-e3.
- Sizar O, Rahman S, Sundareshan V. Amikacin. StatPearls [Internet]: StatPearls Publishing; 2023.
- Korang SK, Safi S, Nava C, Gordon A, Gupta M, Greisen G, et al. Antibiotic regimens for early‐onset neonatal sepsis. Cochrane Database of Systematic Reviews. 2021 (5).
- Korang SK, Safi S, Nava C, Greisen G, Gupta M, Lausten-Thomsen U, et al. Antibiotic regimens for late‐onset neonatal sepsis. Cochrane Database of Systematic Reviews. 2021 (5).
- JAYESH VARIA RP, VANDANA M DESAI, VASAV D DESAI. Adverse Drug Reaction following Injection of Amikacin Sulphate in a Cluster of Preterm Newborns. Indian Journal of Neonatal Medicine and Research. 2017;5(4):NS01-NS3.
- Renna G, Siro-Brigiani G, Cuomo V. Comparative evaluation of the neuromuscular blocking activity of three new aminoglycoside antibiotics in rats. Toxicology letters. 1981;9(2):107-12.
- Potter JM, Edeson R, Campbell R, Forbes A. Potentiation by gentamicin of non-depolarizing neuromuscular block in the cat. Anaesthesia and Intensive Care. 1980;8(1):20-5.
- Regazzi M, Berardi A, Picone S, Tzialla C. Pharmacokinetic and pharmacodynamic considerations of antibiotic use in neonates. Antibiotics. 2023;12(12):1747.
- Simeoli R, Cairoli S, Decembrino N, Campi F, Dionisi Vici C, Corona A, et al. Use of antibiotics in preterm newborns. Antibiotics. 2022;11(9):1142.
- Butranova OI, Ushkalova EA, Zyryanov SK, Chenkurov MS. Developmental pharmacokinetics of antibiotics used in neonatal ICU: focus on preterm infants. Biomedicines. 2023;11(3):940.
- Vella-Brincat JW, Begg EJ, Robertshawe BJ, Lynn AM, Borrie TL, Darlow BA. Are gentamicin and/or vancomycin associated with ototoxicity in the neonate? A retrospective audit. Neonatology. 2011;100(2):186-93.
- Iosifidis E, Evdoridou I, Agakidou E, Chochliourou E, Protonotariou E, Karakoula K, et al. Vancomycin-resistant Enterococcus outbreak in a neonatal intensive care unit: epidemiology, molecular analysis and risk factors. American journal of infection control. 2013;41(10):857-61.
- Clock SA, Ferng Y-H, Tabibi S, Alba L, Patel SJ, Jia H, et al. Colonization with antimicrobial-resistant gram-negative bacilli at neonatal intensive care unit discharge. Journal of the Pediatric Infectious Diseases Society. 2017;6(3):219-26.
- Hasvold J, Bradford L, Nelson C, Harrison C, Attar M, Stillwell T. Gentamicin resistance among Escherichia coli strains isolated in neonatal sepsis. Journal of neonatal-perinatal medicine. 2013;6(2):173-7.
- Zhao R, Yu K, Zhang J, Zhang G, Huang J, Ma L, et al. Deciphering the mobility and bacterial hosts of antibiotic resistance genes under antibiotic selection pressure by metagenomic assembly and binning approaches. Water research. 2020;186:116318.
- Friedland I, Funk E, Khoosal M, Klugman K. Increased resistance to amikacin in a neonatal unit following intensive amikacin usage. Antimicrobial agents and chemotherapy. 1992;36(8):1596-600.
- Rodrigues D, Baldissera GS, Mathos D, Sartori A, Zavascki AP, Rigatto MH. Amikacin for the treatment of carbapenem-resistant Klebsiella pneumoniae infections: clinical efficacy and toxicity. Brazilian Journal of Microbiology. 2021;52(4):1913-9.
- HU W, YANG Q, WANG X, TAN B, CHEN Y, SUN H. Amikacin therapy and management of premature infants infected with carbapenem-resistant Klebsiella pneumoniae. Chinese Journal of Clinical Pharmacology and Therapeutics. 2024;29(12):1401.
- Bor M, Ilhan O. Carbapenem-resistant Klebsiella pneumoniae outbreak in a neonatal intensive care unit: risk factors for mortality. Journal of Tropical Pediatrics. 2021;67(3):fmaa057.
- El Desoky E, Sheikh A, Al Hammadi A. Aminoglycoside and vancomycin serum concentration monitoring and mortality due to neonatal sepsis in Saudi Arabia. Journal of clinical pharmacy and therapeutics. 2003;28(6):479-83.
- Hughes KM, Johnson PN, Anderson MP, Sekar KC, Welliver RC, Miller JL. Comparison of amikacin pharmacokinetics in neonates following implementation of a new dosage protocol. The Journal of Pediatric Pharmacology and Therapeutics. 2017;22(1):33-
- Siddiqi A, Khan D, Khan F, Razzaq A. Therapeutic drug monitoring of amikacin in preterm and term infants. Singapore medical journal. 2009;50(5):486.
- Clark RH, Bloom BT, Spitzer AR, Gerstmann DR. Empiric use of ampicillin and cefotaxime, compared with ampicillin and gentamicin, for neonates at risk for sepsis is associated with an increased risk of neonatal death. Pediatrics. 2006;117(1):67-74.
- Millar M, MacKay P, Levene M, Langdale V, Martin C. Enterobacteriaceae and neonatal necrotising enterocolitis. Archives of disease in childhood. 1992;67(1 Spec No):53-6.
- Furuno JP, Perencevich EN, Johnson JA, Wright M-O, McGregor JC, Morris Jr JG, et al. Methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci co-colonization. Emerging infectious diseases. 2005;11(10):1539.
- Weinstein RA, Fridkin SK. Vancomycin-intermediate and-resistant Staphylococcus aureus: what the infectious disease specialist needs to know. Clinical Infectious Diseases. 2001;32(1):108-15.
- Dua’A A, Xu S, Luig M, Kim HY, Alffenaar J-W. Dosing of vancomycin and target attainment in neonates: a systematic review. International Journal of Antimicrobial Agents. 2022;59(2):106515.
- Kato H, Hagihara M, Okudaira M, Asai N, Koizumi Y, Yamagishi Y, et al. Systematic review and meta-analysis to explore optimal therapeutic range of vancomycin trough level for infected paediatric patients with Gram-positive pathogens to reduce mortality and nephrotoxicity risk. International Journal of Antimicrobial Agents. 2021;58(2):106393.
- Lignieres G, Rybak A, Levy C, Birgy A, Bechet S, Bonacorsi S, et al. Meningitis caused by extended-spectrum β-lactamaseproducing Escherichia coli in infants in France: a case series. JAC-Antimicrobial Resistance. 2023;5(2):dlad042.
