Antibiotic prescribing and resistance patterns among patients admitted to tertiary care hospital with Urinary tract infections at United Arab Emirates (UAE)
Rawda Hassan, Yelly Oktavia Sari , Syed Wasif Gillani , Hassaan Anwer Rathore
Rawda Hassan. College of Pharmacy, Gulf Medical University, Ajman, UAE.
Yelly Oktavia Sari. Department of Pharmacology and Clinical Pharmacy, Faculty of Pharmacy, Universitas Andalas, Padang, Indonesia, yelly.sari@gmail.com
Syed Wasif Gillani*. College of Pharmacy, Gulf Medical University, Ajman, UAE, dr.syedwasif@gmu.ac.ae
Hassaan Anwer Rathore. College of Pharmacy, QU Health, Qatar University, Doha, 2713, Qatar, hrathore@qu.edu.qa
DOI: 10.18549/PharmPract.2026.1.3309
Abstract
Background: Urinary tract infections (UTIs) are among the most common bacterial infections, affecting individuals across all age groups. Gram-negative bacilli, especially E. coli, account for over 80% of acute UTI cases, while other pathogens like Staphylococcus saprophyticus, Klebsiella, Proteus, Pseudomonas, and Enterobacter are less common. Objectives: This study aimed to investigate the epidemiology of uropathogens and assess antimicrobial stewardship practices in the management of urinary tract infections (UTIs) at a tertiary care hospital. Methods: This retrospective study evaluated all adult patients diagnosed with UTIs over a defined time period. Microbiological data was collected to determine the prevalence of uropathogen species. Antimicrobial utilization patterns were analyzed, focusing on initial empiric use of broad-spectrum carbapenems (meropenem) and subsequent de-escalation to more targeted therapy. Results: A total of 124 UTI cases were included in this study. The Escherichia coli was the most prevalent uropathogen, isolated in the majority of UTI cases. Female patients exhibited a higher incidence of UTIs compared to male patients. Initial empiric use of meropenem, was observed in 31.4% of cases. De-escalation of antimicrobial therapy was achieved in 62.1% of cases. A high prevalence (12/16 cases – 75%) of ESBL-producing E. coli was found, which influences empiric therapy choices and de-escalation opportunities. Conclusion: The high prevalence of ESBL-producing E. coli and the relatively high rate of broad-spectrum antibiotic prescribing demonstrated the need for continued antimicrobial stewardship efforts to optimize empiric therapy and promote de-escalation strategies.
Keywords: E. coli, De-escalation, ESBL, Antibiotics, UTI, Broad spectrum
INTRODUCTION
Urinary tract infections (UTIs) are the most common bacterial infections affecting people of all ages1. Failure to diagnose and treat UTIs promptly can result in serious complications, including urinary tract disorders, kidney scarring, high blood pressure, uraemia, and, in pregnant women, an increased risk of premature birth and miscarriage2. Hospital-acquired UTIs, such as cystitis and pyelonephritis, are prevalent, with Escherichia coli (E. coli) being the primary pathogen, causing nearly 80% of infections and affecting 8–10 million people annually in the USA3,4.Globally, the annual cost of treating hospital infections is estimated at 17–29 billion dollars, with 39% of these costs attributed to urinary infections4. Gramnegative bacilli, especially E. coli, are the predominant causes of UTIs, accounting for over 80% of acute cases. Staphylococcus saprophyticus contributes to 5–10% of UTIs, while other bacteria such as Klebsiella, Proteus, Pseudomonas, and Enterobacter are less common and usually associated with urinary system abnormalities or catheter use5,6. Despite the significant reduction in mortality achieved with antibiotics, infectious diseases continue to pose a serious health threat due to the uncontrolled use of antibiotics and the emergence of resistance. Global action is urgently needed to change the consumption and prescription patterns of antibiotics to address the growing resistance crisis7,8. Without a shift in consumption practices, even the development of new medicines may not be sufficient to combat antibiotic resistance. In addition to curbing the unregulated use of antibiotics, preventive measures such as routine vaccination, proper hand hygiene, and attention to food safety are crucial for reducing infection spread9. It’s important to note that antibiotics are effective only against bacterial infections and have no impact on viral infections like colds, sore throats, and influenza. In summary, antibiotic resistance arises through gene mutations, giving rise to new generations of bacteria that are resistant to traditional antibiotic treatments10.
Understanding individual and population-specific risk factors associated with recurrent urinary tract infections (UTIs) can help physicians tailor prophylactic strategies. The narrative review draws on insights from the second Foro en Infections Urticarias Recurrences (FIUR2) symposium, a key Latin American forum dedicated to examining the latest trends and challenges in the treatment of recurrent urinary tract infections (UTIs)10. The information presented is based on a non-systematic review of current literature and the expertise of the forum’s authors and presenters10. Risk factors for UTIs can be classified as behavioural, anatomical, or genetic, and they may vary according to the population being considered and the specific type of UTI. Certain transient conditions, such as pregnancy, can predispose individuals to UTIs or heighten the risk of serious complications from infections. In contrast, permanent conditions like neurogenic bladder dysfunction, often resulting from spinal cord injuries, present a dynamic risk profile for UTIs that evolves over time in response to the changing needs of the patient and the medical interventions they receive11. Controlling for modifiable risk factors, such as through improved hygiene practices or avoiding the use of catheters, can significantly reduce the risk of UTIs. In situations where risk factors are nonmodifiable, the use of prophylactic therapies may be recommended to manage and mitigate the risk of recurrent infections (. Recurrent urinary tract infections (UTIs) are defined as having more than two symptomatic episodes within 6 months or more than three within 12 months. Identifying risk factors is crucial for developing effective prophylactic strategies to reduce the likelihood of recurrence. Additionally. One of the most effective ways to decrease the antibiotics resistance is ADE, antimicrobial deescalation (ADE) is an important strategy in antimicrobial stewardship programs (ASPs) aimed at reducing the use of high-class antibiotics, which are often prescribed initially to effectively treat infectious diseases12. The primary goals of ADE include curbing the emergence of antibiotic-resistant bacteria and striking a balance between providing adequate treatment and avoiding unnecessary antibiotic use in this study we will evaluate the pattern of antibiotic resistance of urinary tract infection, and we will discover the de-escalation contribution to minimize the readmission. The aim of the study was to evaluate the pattern of antibiotic prescribing and resistance patterns among patients admitted to the tertiary care hospital at Ajman, UAE.
MATERIALS AND METHODS
Study Design and duration
The retrospective observational study design was used to evaluate the objectives of this study. The study was conducted overa period of one year (2024).
Study setting
The tertiary care hospital contains diverse clinical environment supports extensive research activities, helping to meet the study’s specific goals and provide valuable insights. The capabilities and environment of the hospital made it an ideal setting for this in-depth study, facilitating a detailed investigation into the impact of antibiotic resistance.
Study population
The study will encompass patients admitted to tertiary care hospital who undergo urine tests and meet the specified inclusion criteria.
Participants Criteria
Inclusion criteria
Patients Admitted with UTI: Inclusion criteria involve patients above 18 who have been diagnosed with urinary tract infection (UTI), and their culture and sensitivity reports are available for analysis.
Exclusion criteria
Cancer and Immunological Diseases: Patients diagnosed with cancer or immunological diseases will not be included in the study.
Patients with Unavailable UTI Reports: Individuals admitted with urinary tract infections (UTIs) but whose culture and sensitivity reports are unavailable will not also be considered for inclusion in the study.
Paediatrics
Sample size calculation
The universal sampling technique was used to collect the data. All the patients who were admitted to hospital and diagnosed with UTI between January 2021 and December 2023 based on inclusion and exclusion criteria were included in the study.
Study procedures
The field investigator underwent specific training for data extraction procedures and was regularly monitored by the research supervisor to maintain data completeness. The study utilized the International Classification of Diseases (ICD-10) diagnostic codes to export and identify UTI-related patient data from various hospital departments, including urology, obstetrics and gynecology, and nephrology. The ICD-10 coding system, introduced by the World Health Organization in 1992, provides a standardized, alphanumeric method for recording and identifying diseases, with each code offering progressively specific diagnostic details. Specific UTI-related codes such as N30.00, N30.21, N30.80, and others were used to access patient records.
Data collection involved a meticulous process of reviewing medical records, with patient information entered into a validated data collection form after a pilot validation of 10 medical reports. All the raw data is stored in with the principal investigator and validated by the IT department prior to analysis. The data validation process includes two independent investigators; randomnly choose patient reports from the raw data and countervalidate with the HMIS of the hospital. Any discrimination more than 15% was resolved with principal investigator.
Ethical Approval
The research project received official approval from the Institutional Review Board (IRB). The study protocol adhers with the ethical guidelines and safeguards to protect the rights and welfare of the participants involved in the research as per the declaration of helsinki.
Data Collection
A specific data collection form was designed according to the Hospital information management system (HMIS) to collect data from patient medication records that are available at clinical pharmacy station computer system.
Primary and Secondary Variables
The data included patient demographic information such as: age, weight, race, gender, and Moreover, the diagnosis upon admission, and a complete medication profile that includes days of hospitalization, dose of antibiotic, de-escalation statues , days of antibiotic therapy, and sensitivity reports Also, it included follow-up data of the re-admission rate of the participants with 30 days of their last admission to be evaluated for the clinical outcome.
Data collection procedure
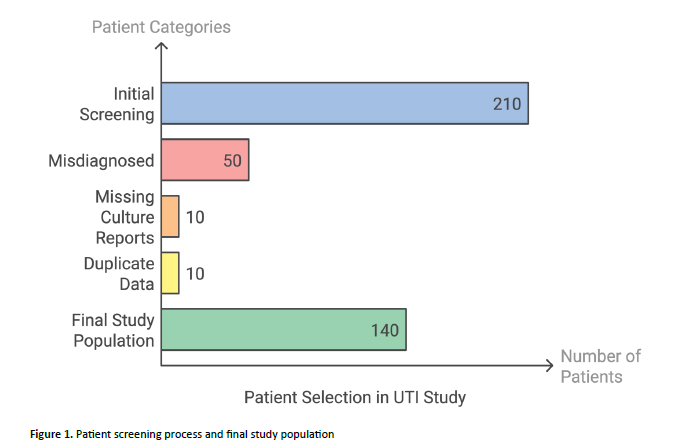
The data collected was entered into Excel file for further processing and data cleaning. There were frequent meetings among investigators (minimum once per month) to go through the data collected. The study initially screened a total of 210 patients diagnosed with Urinary Tract Infection (UTI) but underwent a rigorous selection process to ensure data quality. Of the initial cohort, 50 patients were misdiagnosed with UTI, 10 had missing culture reports, and 10 were identified as having repeated or duplicate data. After applying these exclusion criteria, the final study population consisted of 140 patients with confirmed UTI and complete culture reports Figure 1. This methodical screening process is crucial in clinical research, as it helps eliminate potential sources of bias and ensures that the analysed dataset is both accurate and representative.
Data confidentiality
Several precautions were taken (patients’ names were anonymized and hospital numbers were used instead) to secure patient privacy. Authorized login and password were created to the researcher to avoid any unauthorised access to the data to guarantee data confidentiality. The Google Form was used to securely record all the obtained data, which was then transferred to an Excel file for analysis. However, Ministry of Health and Prevention (MOHAP) or other partner hospitals may request the use of the data only for educational purposes, publications, or other specific purposes. To safeguard the participants’ privacy, patient identifiers won’t be disclosed to any outside parties.
Statistical Analysis
The structured data obtained from Excel was then analysed using the IBM SPSS Statistics (Version 22.0). The analysed data was kept with the principal investigator for five years. The Data was analyzed using both descriptive and inferential statistics. For continous vaariables means ± standard deviation and medians and ranges, were used. Numerical variables were compared for association with chi-square test. Arithmetic means and standard deviations describe the study data .A p-value of < 0.05 was used to determine the statistical significance.
RESULTS
Demographic data of the study population
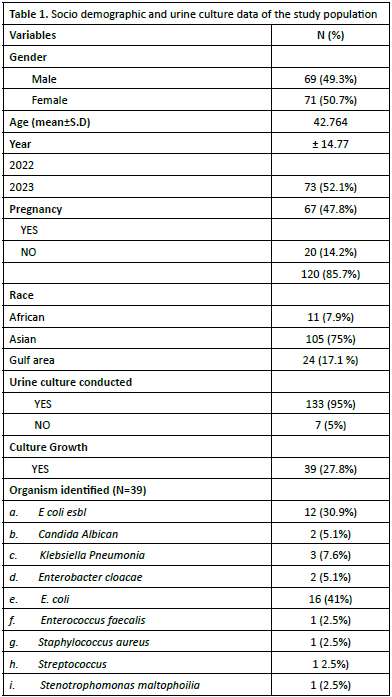
A total of 140 patients were enrolled in this study. The sample comprised 49.3% males and 50.7% females, with a mean age of 42.76±14.77 years. The temporal distribution of patients spanned two years: 2022 (n=73, 52.1%) and 2023 (n=67, 47.8%). Regarding pregnancy status, 14.2% (n=20) of patients were pregnant, while 85.7% (n=120) were non-pregnant. The
racial demographics indicated that Asians constituted the majority (n=105, 75.0%), followed by Gulf region patients (n=24, 17.1%) and Africans (n=11, 7.9%) (Table 1).
Urine Culture and Organism patterns
A total of 140 participants were enrolled in this study, of whom 95% (n=133) underwent urine culture testing, while 5% (n=7) were not tested. Culture positivity was observed in 27.8% (n=39) of tested samples, whereas 72.1% (n=101) demonstrated no microbial growth. Microbiological analysis of positive cultures revealed that Escherichia coli was the predominant Uropathogen, accounting for 41% (n=16) of isolates. Extendedspectrum beta-lactamase (ESBL)-producing E. coli constituted 30.9% (n=12) of cases. Notably, E. coli strains (including ESBLproducing variants) were responsible for 71.9% of all positive cultures, indicating their predominance in urinary tract infections within this study (Table 1).
Clinical Data of the study population
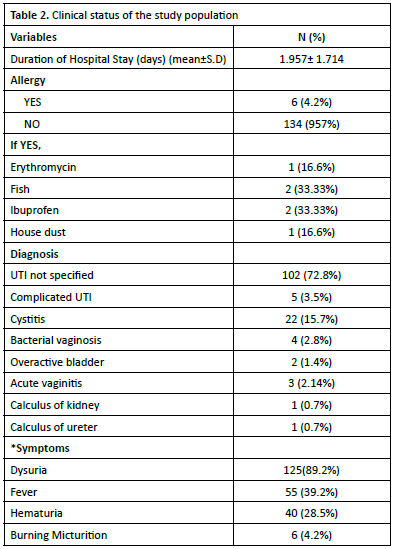
The mean duration of hospitalization was 1.96±1.71 days. Analysis of patient medical histories revealed that 4.2% (n=6) reported allergies, with the following distribution: erythromycin (16.6%, n=1), fish (33.33%, n=2), ibuprofen (33.33%, n=2), and house dust (16.6%, n=1). The majority of participants (95.7%, n=134) reported no known allergies. Clinical manifestations included dysuria as the most frequent symptom (89.2%, n=125), followed by fever (39.2%, n=55), haematuria (28.5%, n=40), and burning micturition (4.2%, n=6). All the details have been shown in Table 2.
Readmission patterns
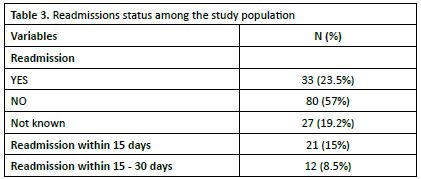
The hospital readmission patterns revealed that 23.5% (n=33) of patients experienced readmission, while 57% (n=80) had no subsequent readmissions. Readmission status was undocumented in 19.2% (n=27) of cases. Among readmitted patients, 15% (n=21) presented within 15 days post-discharge, and 8.5% (n=12) were readmitted between 15-30 days after initial discharge. The data is shown in Table 3.
Microorganism sensitivity and resistance patterns
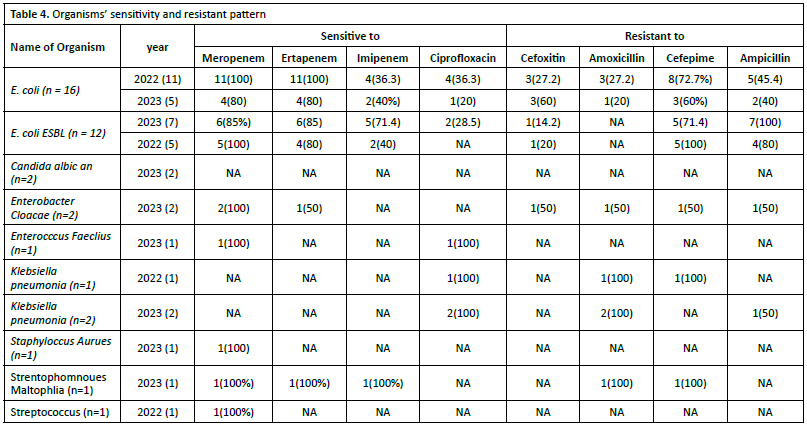
Analysis of antimicrobial susceptibility patterns across 2022- 2023 revealed distinct sensitivity profiles among isolated Ur pathogens. Escherichia coli demonstrated high susceptibility to carbapenems, specifically meropenem (100%) and ertapenem (80-100%), while maintaining moderate sensitivity to imipenem. However, marked resistance was observed against amoxicillin. Similarly, extended-spectrum beta-lactamase (ESBL)-producing E. coli strains exhibited high susceptibility to meropenem and ertapenem (85-100%) but demonstrated resistance to cefepime and ampicillin. Details are provided in Table 4.
Antibiotic Prescribing Patterns
The majority of patients, specifically 76% (106 out of 140), were treated with monotherapy—utilizing a single antimicrobial agent. Conversely, a smaller proportion of patients, approximately 24% (34 out of 140), received combination therapy, which involves using multiple antimicrobial agents simultaneously.
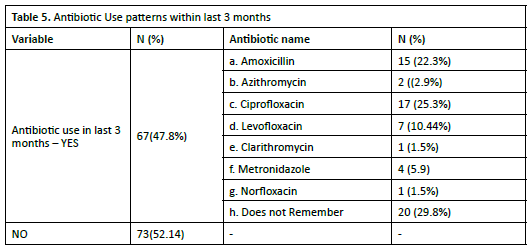
It was also found that (47.8%) of patients reported having used antibiotics within the past three months, while 52.1% did not. Among the patients who utilized antibiotics, a significant proportion (29.8%) reported that they could not recall the specific antibiotic taken (Table 5).
A detailed analysis of antibiotic prescribing practices and usage patterns within a clinical setting involving 140 patients upon admission The findings indicate that the most commonly prescribed antibiotics included meropenem, ciprofloxacin, ceftriaxone, and imipenem with cilastatin, which collectively accounted for approximately 65% of all prescriptions. In assessing the appropriateness of antibiotic prescriptions, it was observed that 109 out of 120 cases (90.8%) were deemed correctly prescribed. Conversely, 6 cases (5%) were categorized as incorrect, while 5 cases (4.2%) were classified as not applicable. Notably, meropenem was the most frequently utilized antibiotic, appearing in both correctly (38.5%) and incorrectly (33.33%) prescribed cases. De-escalation practices were also analysed revealing that 62.1% of patients underwent de-escalation, characterized by a narrowing of their antibiotic regimen based on clinical improvement or specific culture results. Meropenem was the antibiotic most frequently subject to de-escalation, typically switched to ertapenem, followed by ciprofloxacin and ceftriaxone. This reflects a strategic approach towards more targeted therapies following initial broadspectrum coverage.
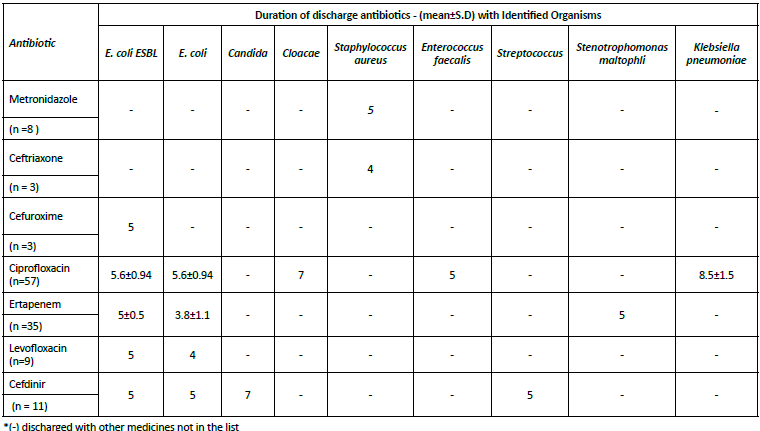
The continuation of antibiotic therapy post-discharge, 128 out of 140 patients (91.4%) maintained treatment, primarily with oral antibiotics. Ciprofloxacin was the most commonly prescribed antibiotic for discharge, indicating a trend towards outpatient-friendly oral therapies, particularly for patients transitioning from hospital to home care. In terms of organismspecific antibiotic use, 39 cases involved identified pathogens, with E. coli—especially extended-spectrum beta-lactamase (ESBL)-producing strains (Table 6).
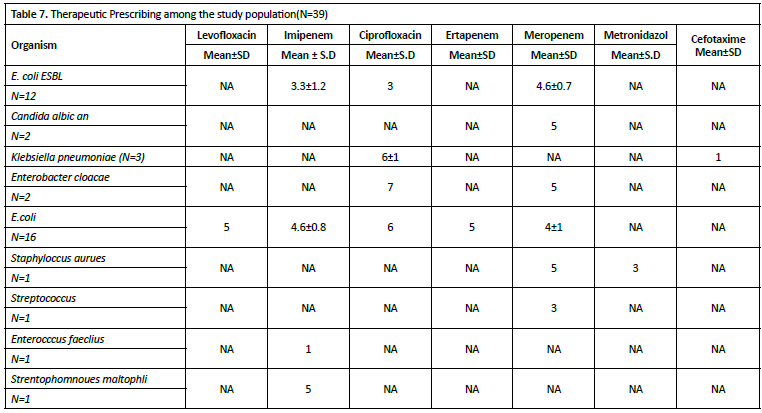
The duration of antimicrobial therapy showed notable variations across different organisms and agents. For ESBLproducing E. coli, treatment durations ranged from 3 days with ciprofloxacin to 4.6±0.7 days with meropenem, while imipenem was administered for 3.3±1.2 days. Non-ESBL E. coli cases demonstrated longer treatment durations, with ciprofloxacin prescribed for 6 days, levofloxacin and ertapenem for 5 days each, imipenem for 4.6±0.8 days, and meropenem for 4±1 days. Klebsiella pneumoniae infections were treated with relatively longer ciprofloxacin courses (6±1 days), while also receiving shorter cefotaxime therapy (1 day) (Table 7).
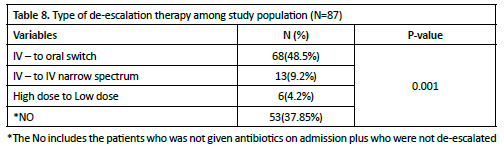
The Table 8 – 9 presents the usage patterns of antibiotic treatment de-escalation patterns, highlighting three categories: IV to oral switch, IV to IV narrow spectrum, and high dose to low dose. The largest group was the IV to oral switch, with 68 (48.5%) patients, followed by the IV-to-IV narrow spectrum with 13 (9.2%) patients. The transition from high dose to low dose was minimal, with only 4 (4.2%) patients.
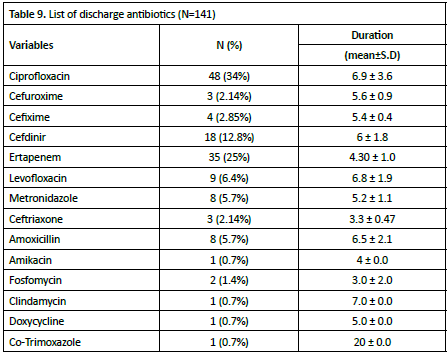
The analysis of discharge antibiotics comparison with identified organism has been conducted. Ciprofloxacin had the highest usage (n=57, 45.2% of total prescriptions), Ertapenem was the second most prescribed (n=35, 27.8%), Cefdinir (n=11, 8.7%), Levofloxacin (n=9, 7.1%), Metronidazole (n=8, 6.3%), ceftriaxone (n=3, 2.4%) and Cefuroxime (n=3, 2.4%). Ciprofloxacin was the most consistently prescribed around 5.6 days for E. coli (both ESBL and non-ESBL strains), longer duration for Cloacae (7 days) and K. pneumoniae (8.5±1.5 days). Standard deviation of 0.94 days suggests relatively consistent dosing practices. Ertapenem has variable duration between ESBL and non-ESBL, E. coli: 5±0.5 days and Non-ESBL E. coli: 3.8±1.1 days. Also prescribed for S. maltophilia (5 days). Cefdinir was Consistent 5-day duration for E. coli strains. It has Longer duration (7 days) for Candida infections (Table 10).
Clinical Outcomes
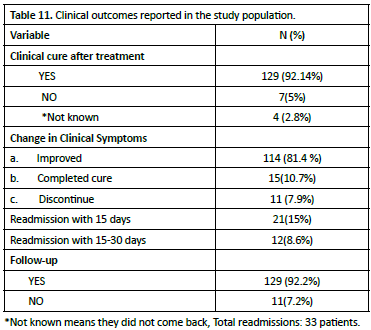
A total of 114 (81.4%) patients showed clinical improvment, while complete cure was achieved in 15 (10.7%) patients. Treatment discontinuation was observed in 11 (7.9%) cases. In terms of hospital readmissions, 21 (15.0%) patients required readmission during the first 15 days following treatment, while an additional 12 (8.5%) patients were readmitted between days 15 and 30, resulting in a total readmission rate of 33 (23.5%) patients. Patient follow-up compliance was notably high, with 129 (92.2%) patients attending their scheduled follow-up appointments, while follow-up was not completed by 11 (7.2%) patients (Table 11).
Association between Clinical Outcomes with De-escalation Therapy
In terms of de-escalation therapy, 37.9% (49 patients) received this approach, while 62.1% (80 patients) did not undergo deescalation. Symptom changes showed similar patterns: among 114 patients with improved symptoms, 37.7% were associated with de-escalation, while 62.3% were not. For the 15 patients with complete cure, 40% were linked to de-escalation therapy. Readmission rates varied by time frame: within 15 days, only 10% of patients were readmitted, while in the 15-30 day window, 41.6% of patients experienced readmission (Table 12).
DISCUSSION AND LIMITATIONS
Demographic data of the study population
The demographic profile of our study population reveals
several noteworthy characteristics and potential implications for the interpretation of our findings. The study achieved a wellbalanced gender distribution, with nearly equal representation of males (49.3%) and females (50.7%), which enhances the generalizability of our results across genders and minimizes potential gender-related bias in our findings. The mean age of 42.76 ± 14.77 years indicates a predominantly middle-aged study population, with considerable variation as suggested by the standard deviation. This age range is particularly relevant as
it represents the demographically active workforce population, making our findings particularly applicable to occupational health considerations. However, the relatively large standard deviation suggests that our results may be generalizable across different age groups, from young adults to older individuals The racial distribution of our study population presents both strengths and limitations. Asian Predominance: The substantial representation of Asian participants (75.0%) reflects the demographic reality of the study region but may limit the generalizability of our findings to other racial groups. Gulf Region Representation: The inclusion of Gulf region patients (17.1%) provides valuable data for this specific population, which is often underrepresented in international studies.
African Representation: While the African was relatively small (7.9%), their inclusion adds diversity to our sample and provides preliminary data for this demographic group. In Crosssectional study conducted in Saudi Arabia, particularly in Jazan Province, located in the southwestern region. A total of 1082 urinary bacterial samples were isolated and identified. Females accounted for more bacterial infections (62.66%) than males (37.34%) which is similar to our study13 .In cross-sectional study was conducted among consecutively selected outpatients (n = 344) from January to May 2022 at Morogoro Regional Referal Hospital he prevalence of UTIs was 41% (141/344) and elders (>=60 years) had five times higher odds of having UTI as compared to adolescents (p < 0.001)14.
Urine Culture and Organism patterns
The striking predominance of E. coli (total 71.9% including ESBL strains) in our study merits particular attention. The 41% prevalence of non-ESBL E. coli aligns with global patterns of Ur pathogen distribution. The high proportion of ESBLproducing strains (30.9%) is concerning and has significant therapeutic implications. The combined prevalence suggests that empiric therapy should maintain adequate coverage for both ESBL, and non-ESBL E. coli strains Our findings have several important clinical implications. Our study findings have several significant clinical implications that warrant careful consideration in practice. Regarding empiric therapy selection, the high prevalence of both ESBL (30.9%) and non-ESBL E. coli (41%) necessitates thoughtful antibiotic choices guided by local resistance patterns and patient-specific risk factors, including previous antibiotic exposure, recent hospitalizations, and healthcare facility contact. In terms of antimicrobial stewardship, the notable ESBL prevalence calls for targeted interventions, such as selective reporting of susceptibility results, implementation of resistance prevention strategies, and regular monitoring of local resistance patterns through systematic surveillance. The diagnostic approach should maintain the high testing rates (95%) demonstrated in our study, with particular attention to risk stratification for resistant organisms and consideration of fungal cultures in appropriate clinical scenarios such as immunocompromised patients or those with prolonged antibiotic exposure. The integration of these evidence-based approaches into clinical practice, coupled with regular evaluation and updates based on local resistance patterns, will help optimize patient outcomes while promoting responsible antimicrobial use. A cross-sectional study at the departments of General Medicine, Nephrology, and Urology at Nishtar Hospital, Multan, from May 5, 2019, to November 5, 2019.The study showed E. coli to be the most common pathogen in UTI, with very high antibiotic resistance. This warrants the careful selection and conservative use of antibiotics15. Among 53 positive urine cultures, Escherichia coli was detected in 21 (39.6%)15. In 2022, all the 11 E. coli samples in our study were sensitive to Meropenem and Ertapenem, while sensitivity to Imipenem was 36.3%. Resistance was noted in 72.7% for Cefepime and 45.4%forAmpicillin. In 2023, sensitivity to Meropenem and Ertapenem decreased to 80%, with a notable increase in resistance to Cefoxitin (60%). In 2023, 85% of E. coli esbl were sensitive to Meropenem and Ertapenem, with 71.4% sensitive to Imipenem. Resistance to Amoxicillin was observed in 14.2%. In 2023, both candida samples were 100% sensitive to Meropenem., but only 50% sensitivity to Imipenem. In 2022, Klebsiella pneumoniae was sensitive to ciprofloxacin but, resistant to amoxicillin and ampicillin. In 2023, both samples remained 100% sensitive to Ciprofloxacin but exhibited varying resistance patterns. Staphylococcus aureus in 2023, the single sample was 100% sensitive to Meropenem. Stenotrophomonas melophilia: in 2023, the single sample showed 100% sensitivity to all tested antibiotics. Streptococcus: In 2022, the single sample was 100% sensitive to Meropenem. The p-values indicate the statistical significance of the differences observed between the years for each organism. For example, E. coli showed a p-value of 0.42 in 2022 and 0.4 in 2023, suggesting no significant change in sensitivity patterns over the years. The same study we used to compare with that’s done at Melton hospital has demonstrated the almost same results. Of all these, E. coli cultures, 21 (100.0%) were sensitive to polymyxin B, colistin, and ertapenem, followed by 18 (85.7%) sensitive to amikacin, 15 (71.4%) sensitive to imipenem, 14 (66.7%) sensitive to gentamicin, 13 (61.9%) sensitive to meropenem, nine (42.9%) sensitive to ampicillin, eight (38.1%) sensitive to piperacillin/tazobactam, seven (33.3%) sensitive to cefoperazone/sulbactam, six (28.6%) sensitive to co-amoxiclav, cefotaxime, and ceftriaxone, and five (23.8%) sensitive to cefuroxime and ciprofloxacin. All 21 (100.0%) cultures were resistant to amoxicillin. Enterococcus species were detected in 18 (33.9%) of all the positive cultures. Of these 18 enterococci cultures, five (27.8%) were sensitive to ciprofloxacin. All 18 (15.0%) cultures were resistant to cefotaxime, ceftriaxone, and cefuroxime. Klebsiella species were detected in one (1.9 %) of all the positive cultures. Growth was resistant to amikacin, ampicillin, co-amoxiclav15. In a previous study conducted in patients attending hospitals in Bushenyi district western Uganda by Odoki and others, a significantly high prevalence of E. coli 41.9% was reported16. A study in Duhok City, Kurdistan Region of Iraq has similar results as our results,the E. coli was found to be highly susceptible to ertapenem (97.6%) and imipenem (96.4%) but resistant to ampicillin (87.8%)17. other studies found that 100% of E. coli isolates were sensitive to gentamycin, amikacin, imipenem, meropenem, piperacillintazobactam, and tobramycin18. In study done at the Laboratory of Microbiology, Faculty of Medicine, Universitas Sumatera Utara, Medan, Indonesia , It was having a similar results to our study as a total of 20 K. pneumoniae isolates were obtained in this study. The results showed that K. Pneumonia isolates exhibited good sensitivity to amikacin (100%) and meropenem (80%). Sensitivity of levofloxacin (60%), ceftazidime (55%), ciprofloxacin (55%), cefoperazone (50%), and cotrimoxazole (50%) were moderate for the bacterial isolates. K. Pneumoniae isolates indicated low sensitivity to cefuroxime (45%), chloramphenicol (35%), cefepime (30%), cefotaxime (30%), tetracycline (30%), amoxicillin (5%), and ampicillin (5%)19.
Antibiotic Prescribing Patterns
In our study the analysis reveals a clear preference Mono therapy, with more than three-quarters of patients (76%, n=106) receiving mono antimicrobial agents. In contrast, approximately combination therapy quarter of patients (24%, n=34) were managed with combination, suggesting a predominant clinical practice favouring multi-drug approaches. In Systematic Review and Meta-Analysis Study for Monotherapy versus combination therapy for multidrug-resistant Gram-negative infections that Published: 29 October 2019, the studies included a total of 4514 patients: 41% of patients (n = 1848) were treated with monotherapy, 59% (n = 2666) with combination therapy20
Clinical status of the study population
The mean duration of hospitalization was approximately 1.96 days, with a standard deviation of 1.71 days. A small percentage of participants, specifically 4.2% (n=6), reported having allergies, with the most common allergens being fish and ibuprofen, each affecting 33.33% of those with allergies. The predominant clinical diagnosis among the participants was uncomplicated urinary tract infection (UTI), accounting for 72.8% (n=102) of cases. Other diagnoses included cystitis (15.7%, n=22), complicated UTI (3.5%, n=5), bacterial vaginosis (2.8%, n=4), and acute vaginitis (2.14%,n=3). In terms of clinical manifestations, dysuria was the most frequently reported symptom, observed in 89.2% (n=125) of participants, followed by fever (39.2%, n=55) and hematuria (28.5%, n=40). Burning micturition was reported by 4.2% (n=6) of the participants. Dysuria typically affects approximately 3% of all adults over 40 years of age at any given time, making it the most common urinary symptoms21.
Prescribing Pattern
The analysis also reveals several significant patterns in antibiotic prescribing practices within this clinical setting. The predominant use of broad-spectrum antibiotics, particularly meropenem, ciprofloxacin, ceftriaxone, and imipenem with cilastatin (accounting for 65% of prescriptions), reflects a conservative empirical approach to initial infection management. The high rate of appropriate prescribing (90.8%) suggests strong adherence to clinical guidelines and effective antimicrobial stewardship practices. The substantial de-escalation rate of 62.1% demonstrates a commendable commitment to optimizing antibiotic therapy based on clinical response and culture results, particularly with meropenem being frequently de-escalated to ertapenem. The high continuation rate of antibiotic therapy post-discharge (91.4%), primarily with oral antibiotics like ciprofloxacin, indicates effective transition planning from inpatient to outpatient care. The prevalence of E. coli, especially ESBL-producing strains, as the dominant pathogen (71.8% of identified cases) and the corresponding use of carbapenems highlights the growing challenge of antibiotic-resistant organisms in clinical practice and validates the initial broad-spectrum antibiotic approach. These findings collectively suggest a well-structured antibiotic management program that balances the need for broad initial coverage with appropriate de-escalation strategies, while effectively managing the transition to outpatient care. Most commonly prescribed antibiotics included meropenem, ciprofloxacin, ceftriaxone, and imipenem with cilastatin, which collectively accounted for approximately 65% of all prescriptions. This reliance on broadspectrum broadspectrum antibiotics for initial treatment underscores the typical empirical therapy strategies employed to address a wide range of potential infections. In assessing the appropriateness of antibiotic prescriptions, it was observed that 109 out of 120 cases (90.8%) were deemed correctly prescribed. Conversely, 6 cases (5%) were categorized as incorrect, while 5 cases (4.2%) were classified as not applicable. Notably, meropenem was the most frequently utilized antibiotic, appearing in both correctly (38.5%) and incorrectly (33.33%) prescribed cases. Deescalation practices were also analysed revealing that 62.1% of patients underwent de-escalation, characterized by a narrowing of their antibiotic regimen based on clinical improvement or specific culture results. Meropenem was the antibiotic most frequently subject to de-escalation, typically switched to ertapenem, followed by ciprofloxacin and ceftriaxone. This reflects a strategic approach towards more targeted therapies following initial broad-spectrum coverage. Regarding the continuation of antibiotic therapy post-discharge, 128 out of 140 patients (91.4%) maintained treatment, primarily with oral antibiotics. Ciprofloxacin emerged as the most commonly prescribed antibiotic for discharge, indicating a trend towards outpatient-friendly oral therapies, particularly for patients transitioning from hospital to home care. In terms of organismspecific antibiotic use, 39 cases involved identified pathogens, with E. coli—especially extended-spectrum beta-lactamase (ESBL)-producing strains—being the most prevalent, accounting for 71.8% of cases. Carbapenems such as meropenem and imipenem were predominantly utilized to treat both ESBL and non-ESBL-producing E. coli, underscoring the critical role these antibiotics play in managing severe bacterial infections
Admission antibiotics
Meropenem was the most frequently prescribed empirical antibiotic on admission, administered to 44 patients (31% of cases). Ciprofloxacin was the second most common choice, used in 19 patients (13.5%), followed by ceftriaxone in 15 patients (10.7%). Imipenem/cilastatin was administered to 14 patients (10% of cases). Doxycycline and amoxicillin were the least frequently prescribed antibiotics on admission to 2(1.4%) and 2(1.4%) respectively .In Therapeutics Letter 135 discusses antimicrobial best practices for treating uncomplicated UTIs in British Columbia (BC), Canada recommend Empiric prescription of nitrofurantoin for 5 to 7 days is recommended.[22,23,24] The British Columbia Centre for Disease Control recommends one oral dose of Fosfomycin 3 g when CrCl is ≤ 30mL/min or when there is allergy/intolerance to nitrofurantoin25. In standard Escherichia coli infections, Meropenem was the most frequently prescribed antibiotic, used in 7 cases (43.7%), followed by Imipenem in 5 cases (31.2%), with Ciprofloxacin, Levofloxacin, and Doxycycline used in additional cases. For ESBL-producing E. coli, a similar pattern emerged, with Meropenem leading at 6 cases (50%) and Imipenem following at 4 cases (33.3%). Klebsiella species infections were primarily treated with Ciprofloxacin, accounting for 2 cases (6.6%). In Staphylococcus aureus cases, both Meropenem and Ceftriaxone were employed as the drugs of choice, each used in one case (100%). This data demonstrates a predominant use of carbapenem antibiotics, particularly in E. coli infections, while other pathogens were managed with targeted antibiotic choices. A hospital-based cross-sectional study in Sri Lanka examined antibiotic susceptibility patterns in ESBL-producing organisms causing urinary tract infections, revealing varying levels of effectiveness: meropenem showed the highest efficacy at 95% (58 patients), followed by imipenem at 73.7% (45 patients), amikacin at 60.6% (37 patients), and nitrofurantoin at 45.9% (28 patients). A concerning finding was that three patients (4.9%) developed E. coli resistance to meropenem, all of whom had previously received multiple antibiotics, including meropenem, for recurrent UTIs. This pattern aligns with carbapenem resistance documented in India among 167 patients in 2014, highlighting a growing regional concern. While carbapenems (meropenem and imipenem) remain the most effective treatment options for ESBL-producing organisms, the emergence of resistant strains emphasizes the critical need for careful antibiotic stewardship, regular monitoring of resistance patterns, and consideration of patient treatment history when selecting antibiotics28.
Discharge antibiotics
The analysis of discharge antibiotics reveals distinct patterns in both prescription preferences and treatment durations. Ciprofloxacin emerges as the predominantly prescribed antibiotic, accounting for 34% of cases (48 patients) with a moderate treatment duration of 6.9 ± 3.6 days, suggesting its versatility in treating various infections. Ertapenem follows as the second most common choice (25% of cases, 35 patients) with a shorter duration of 4.30 ± 1.0 days, indicating its potential role in targeted therapy. Notably, Co-Trimoxazole, although prescribed to only one patient, had the longest treatment duration (20.0 ± 0.0 days), suggesting its use in specific conditions requiring extended therapy. The majority of antibiotics show treatment durations clustering around 5-7 days, with Fosfomycin and Ceftriaxone having the shortest durations (3.0 ± 2.0 and 3.3 ± 0.47 days respectively), likely reflecting their use in specific, acute conditions. This variability in treatment durations across different antibiotics likely reflects both the nature of the infections being treated and established clinical protocols for specific pathogens. The predominance of oral antibiotics in the discharge medication list, particularly fluoroquinolones (Ciprofloxacin and Levofloxacin), aligns with the goal of facilitating outpatient management while maintaining therapeutic efficacy. Ciprofloxacin is an antibiotic agent in the fluoroquinolone class used to treat bacterial infections such as urinary tract infections and pneumonia29,30. Levofloxacin is a third-generation of fluoroquinolone drug that is molecularly isomeric for Ofloxacin and not only inherits its broad-spectrum antibacterial characteristics, but also can enhance antibacterial activity on this basis31,32,33. IN metaanalysis, 5 studies (RCTs) were included, involving a total of 2,877 patients. The analysis results showed that in the treatment of UTI diseases, there was no significant difference in the efficacy and safety of levofloxacin and ciprofloxacin34.
Type of de-escalation therapy among study population
The de-escalation patterns observed in this study reveal significant insights into antibiotic management strategies within the clinical setting. The predominant use of IV to oral switch (48.5% of cases) as the primary de-escalation method suggests a strong preference for transitioning patients to oral therapy when clinically appropriate, which aligns with antimicrobial stewardship principles by potentially reducing hospital stays, costs, and complications associated with IV therapy. The relatively lower proportion of IV to IV narrow spectrum transitions (9.2%) indicates more selective use of this approach, possibly reserved for cases where oral therapy is not suitable but a narrower spectrum antibiotic is warranted. The minimal utilization of high dose to low dose adjustments (4.2%) suggests this is not a commonly employed strategy, perhaps reflecting a preference for maintaining therapeutic dosing until treatment completion rather than dose reduction. Notably, the substantial proportion of patients (37.85%) who did not undergo any de-escalation warrants attention and may reflect cases with severe infections, resistant organisms, or other clinical factors necessitating continued broad-spectrum therapy. The statistical significance of these distribution patterns (p=0.001) confirms that these are not random variations but rather reflect structured clinical decision-making processes in antibiotic management. A significant cross-sectional study conducted in critical care units has demonstrated the safety and cost-effectiveness of switching from intravenous (IV) to oral antibiotics. The research revealed no difference in mortality rates between patients who were switched to oral antibiotics and those who continued on IV therapy, confirming the safety of this approach even in critically ill patients. While the overall hospitalization costs remained similar between both groups (p > 0.05), patients who switched to oral antibiotics showed notably lower daily intensive care expenses and reduced antimicrobial treatment costs35. These findings strongly support implementing IV-to-oral antibiotic conversion as a safe and economically beneficial strategy in critical care settings, provided patients meet the clinical criteria for the switch. This practice represents an important advancement in antibiotic stewardship, offering a way to reduce healthcare costs without compromising patient safety or treatment outcomes36.
Association between Clinical outcomes and De-escalation therapy
The retrospective analysis of this clinical trial data suggests that the de-escalation therapy intervention did not have a statistically significant impact on primary patient outcomes. Rates of clinical cure, symptomatic improvements, and treatment discontinuation were comparable between the deescalation therapy cohort and the non-therapy control group, indicating that factors beyond the de-escalation approach were likely more influential in driving these clinical endpoints. However, the data did demonstrate a potential short-term benefit, as patients receiving de-escalation therapy exhibited notably lower readmission rates within the initial 15-day posttreatment period compared to the non-therapy group. Yet this advantage appeared to wane over the longer 15–30-day follow-up window, with readmission rates converging between the two cohorts. Overall, the relatively minor magnitude of differences observed between the therapy and control groups implies that the de-escalation intervention alone was not a primary determinant of the patient outcomes analysed in this study, suggesting the need for further research to elucidate the optimal role, if any, of de-escalation strategies within this clinical context. A study of 473 intensive care unit (ICU) patients with infectious diseases between 2018-2020 compared outcomes across three groups: ADE (53 patients, 11.2%), No Change (173 patients, 36.6%), and Other Change (247 patients, 52.2%). The 14-day death rates were 9.4% in the ADE group, 11.6% in the No Change group, and 21.9% in the Other Change group. After adjusting for other factors that might influence survival, the No Change group had a significantly higher risk of death (56% higher) compared to the ADE group (p = 0.0181). While the Other Change group showed a 28% higher risk of death compared to the ADE group, this difference was not statistically significant (p = 0.1874), meaning it could have occurred by chance[37]. In retrospective cohort observational study conducted over one year at Prince Sultan Military Medical City (PSMMC), ninety-one patients were enrolled and analysed. The study found that while baseline characteristics were comparable between groups, only 29.7% (27 patients) achieved successful de-escalation, while the majority 70.3% (64 patients) failed to experience de-escalation. The findings demonstrated that successful de-escalation was associated with improved patient outcomes, specifically in terms of reduced hospital length of stay (LOS), though the specific patient admission criteria were not specified in the given information38. A reduction in hospital LOS was shown, which was also found in previous studies39,40.
CONCLUSION
The study concluded high rate of broad-spectrum antibiotic prescribing and this may reflect local resistance patterns, warranting ongoing antimicrobial stewardship attention.Deescalation of antimicrobial therapy was achieved in 62.1% of cases, indicating good stewardship practices. A high prevalence of extended-spectrum beta-lactamase (ESBL)-producing E. coli was observed, which influences empiric therapy choices and de-escalation opportunities.
References
- Medina M, Castillo-Pino E. An introduction to the epidemiology and burden of urinary tract infections. Ther Adv Urol.2019;11:1756287219832172.
- Uwaezuoke SN, Ndu IK, Eze IC. The prevalence and risk of urinary tract infection in malnourished children: a systematic review and meta-analysis. BMC Pediatr. 2019;19(1):1-20.
- Abbo LM, Hooton TM. Antimicrobial stewardship and urinary tract infections. Antibiotics (Basel). 2014;3(2):174-92.
- Foxman B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Dis Mon. 2003;49(2):53-70.
- European Medicines Agency. Trends in the sales of veterinary antimicrobial agents in nine European countries (2005–2009). London: European Medicines Agency; 2011.
- Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol. 2015;13(5):269-84.
- Mortazavi F, Shahin N. Changing patterns in sensitivity of bacterial uropathogens to antibiotics in children. Pak J Med Sci. 2009;25(5):801-5.
- Kibret M, Abera B. Antimicrobial susceptibility patterns of E. coli from clinical sources in northeast Ethiopia. Afr Health Sci. 2011;11:40-5.
- Raj JRM, Vittal R, Shivakumaraswamy SK, Deekshit VK, Chakraborty A, Karunasagar I. Presence & mobility of antimicrobial resistance in Gram-negative bacteria from environmental samples in coastal Karnataka, India. Indian J Med Res. 2019;149(2):290.
- Davies J, Davies D. Origins and evolution of antibiotic resistance. Microbiol Mol Biol Rev. 2010;74(3):417-33.
- NCBI Bookshelf. Available from: www.ncbi.nlm.nih.gov/books/NBK560617/. Accessed 1 Nov 2024.
- Zhao K. Effect of antimicrobial de-escalation strategy on 14-day mortality among intensive care unit patients: a retrospective propensity score-matched cohort study with inverse probability-of-treatment weighting. BMC Infect Dis. 2023;23:8491.doi:10.1186/s12879-023-08491-7.
- Alhazmi AH, Alameer KM, Abuageelah BM, Alharbi RH, Mobarki M, Musawi S, et al. Epidemiology and antimicrobial resistance patterns of urinary tract infections: a cross-sectional study from southwestern Saudi Arabia. Medicina (Kaunas). 2023;59(8):1411.
- Mlugu EM, Mohamedi JA, Sangeda RZ, et al. Prevalence of urinary tract infection and antimicrobial resistance patterns of uropathogens with biofilm forming capacity among outpatients in Morogoro, Tanzania: a cross-sectional study. BMC Infect Dis. 2023;23:660.
- Zammil M, Adnan M, Sikandar SM, Waheed MU, Javed N, Ur Rehman MF. Study of culture and sensitivity patterns of urinary tract infections in patients presenting with urinary symptoms in a tertiary care hospital. Cureus. 2020;12(2):e7013.
- Odoki M, Aliero AA, Tibyangye J, et al. Prevalence of bacterial urinary tract infections and associated factors among patients attending hospitals in Bushenyi district, Uganda. Int J Microbiol. 2019;2019:4246780.
- Naqid IA, Balatay AA, Hussein NR, Saeed KA, Ahmed HA, et al. Antibiotic susceptibility pattern of Escherichia coli isolated from various clinical samples in Duhok City, Kurdistan Region of Iraq. Int J Infect. 2020;7(3):e103740.
- Akter T, Hossain M, Khan M, Sultana H, Fatema K, Al Sanjee S, et al. Isolation, identification and antimicrobial susceptibility pattern analysis of Escherichia coli isolated from clinical samples of Bangladesh. Asian J Biomed Pharm Sci. 2016;6:13-6.
- Schmid A, Wolfensberger A, Nemeth J, et al. Monotherapy versus combination therapy for multidrug-resistant Gram-negative infections: systematic review and meta-analysis. Sci Rep. 2019;9:15290.
- Michels TC, Sands JE. Dysuria: evaluation and differential diagnosis in adults. Am Fam Physician. 2015;92(9):778-86.
- American Geriatrics Society 2019 Updated AGS Beers Criteria R for potentially inappropriate medication use in older adults. J Am Geriatr Soc.2019;67(4):674-94. doi:10.1111/jgs.15767.
- Hoang P, Salbu RL. Updated nitrofurantoin recommendations in the elderly: a closer look at the evidence. Consult Pharm. 2016;31(7):381-4. doi:10.4140/TCP.n.2016.381.
- Gardiner BJ, Stewardson AJ, Abbott IJ, Peleg AY. Nitrofurantoin and fosfomycin for resistant urinary tract infections: old drugs for emerging problems. Aust Prescr. 2019;42(1):14-9. doi:10.18773/austprescr.2019.002.
- Ten Doesschate T, van Haren E, Wijma RA, et al. The effectiveness of nitrofurantoin, fosfomycin and trimethoprim for the treatment of cystitis in relation to renal function. Clin Microbiol Infect. 2020;26(10):1355-60. doi:10.1016/j.cmi.2020.03.001.
- Cai T, Tamanini I, Tascini C, et al. Fosfomycin trometamol versus comparator antibiotics for the treatment of acute uncomplicated urinary tract infections in women: a systematic review and meta-analysis. [Journal details incomplete]
- Fernando MMPSC, Luke WANV, Miththinda JKND, et al. Extended spectrum beta lactamase producing organisms causing urinary tract infections in Sri Lanka and their antibiotic susceptibility pattern: a hospital-based cross-sectional study. BMC Infect Dis. 2017;17:138.
- Shaikh S, Fatima J, Shakil S, Rizvi SMD, Kamal MA. Risk factors for acquisition of extended spectrum beta lactamase producing Escherichia coli and Klebsiella pneumoniae in North-Indian hospitals. Saudi J Biol Sci. 2015;22(1):37-41.
- Zhang HL, Tan M, Qiu AM, Tao Z, Wang CH. Antibiotics for treatment of acute exacerbation of chronic obstructive pulmonary disease: a network meta-analysis. BMC Pulm Med. 2017;17(1):196.
- Bartolomé-Álvarez J, Solves-Ferriz V. Increase in methicillin-resistant and ciprofloxacin-susceptible Staphylococcus aureus in osteoarticular, skin and soft tissue infections. Rev Esp Quimioter. 2020;33(2):143-4.
- Dawe RS, Ibbotson SH, Sanderson JB, et al. A randomized controlled trial (volunteer study) of sitafloxacin, enoxacin, levofloxacin and sparfloxacin phototoxicity. Br J Dermatol. 2003;149:1232-41.
- von Gottberg A, Klugman KP, Cohen C, et al. Emergence of levofloxacin-non-susceptible Streptococcus pneumoniae and treatment for multidrug-resistant tuberculosis in children in South Africa: a cohort observational surveillance study. Lancet. 2008;371:1108-13.
- Schaeffer EM. Re: Levofloxacin for BK Virus Prophylaxis following Kidney Transplantation: a randomized clinical trial. J Urol. 2015;194:410.
- Xue Z, Xiang Y, Li Y, Yang Q. A systematic review and meta-analysis of levofloxacin and ciprofloxacin in the treatment of urinary tract infection. Ann Palliat Med. 2021;10(9):9765-71. doi:10.21037/apm-21-2042.
- Gasparetto J, Tuon FF, dos Santos Oliveira D, et al. Intravenous-to-oral antibiotic switch therapy: a cross-sectional study in critical care units. BMC Infect Dis. 2019;19:650. doi:10.1186/s12879-019-4280-0.
- Schuts EC, Hulscher M, Mouton JW, et al. Current evidence on hospital antimicrobial stewardship objectives: a systematic review and meta-analysis. Lancet Infect Dis. 2016;16:847-56.
- Zhao K, Zhang Z, Liang Y, et al. Effect of antimicrobial de-escalation strategy on 14-day mortality among intensive care unit patients: a retrospective propensity score-matched cohort study with inverse probability-of-treatment weighting. BMC Infect Dis. 2023;23:508. doi:10.1186/s12879-023-08491-7.
- Hareef H, Alfahad W, Albaadani A, Alyazid H, Talib RB. Impact of antibiotic de-escalation on hospitalized patients with urinary tract infections: a retrospective cohort single center study. J Infect Public Health. 2020;13(7):985-99.
- Khasawneh FA, Karim A, Mahmood T, Ahmed S, Jaffri SF, Tate ME, et al. Antibiotic de-escalation in bacteremic urinary tract infections: potential opportunities and effect on outcome. Infection. 2014;42(5):829-34.
- Rieger KL, Bosso JA, MacVane SH, Temple Z, Wahlquist A, Bohm N. Intravenous-only or intravenous transitioned to oral antimicrobials for Enterobacteriaceae-associated bacteremic urinary tract infection. Pharmacotherapy. 2017;37(11):1479-83.
- Nadeem S, Hameed AA, Gillani SW, Rathore HA. Reported Postoperative Surgical Site Infections in Tertiary Care Hospitals, Systematic Review of Recent Literature. Curr Drug Saf. 2025 Jan 28. doi: 10.2174/0115748863338553250106111958.
